|
|
Post by dogsbody on May 19, 2010 11:11:46 GMT
Oh of course socold. Everyone is wrong except IPCC, and you. There is Solomon's 1000 year Reich nonsense too. Why was it again there is no necessity for the tropical troposphere "hot spot" despite all GCM's predicting it? Please, no barf mulch from ReinventedClimate. And why isn't the stratosphere cooling as advertised it should? Is there anything at all that is falsifiable? 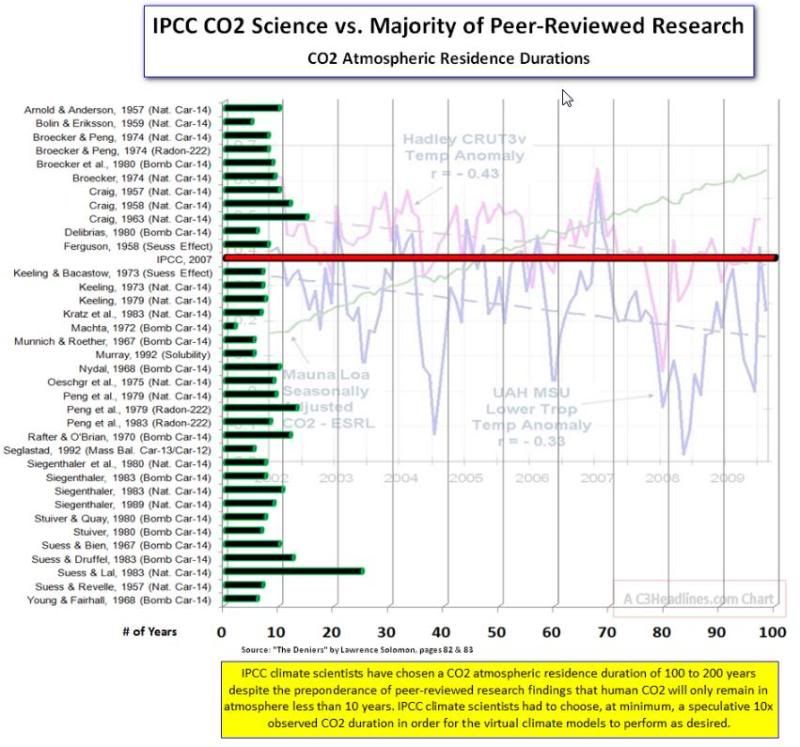 You need to be careful here. I'd like to know eactly what those studies say. Socold's right there is a lot of confusion about this issue. There is a big difference between the turnover time and the time taken to remove 'excess' CO2 from the atmosphere. The turnover time or average life time of a CO2 molecule is ~5 years. This is easy enough to demonstrate. The atmosphere holds about 750 GtC (gigatons of carbon). During the annual cycle ~150 GtC is removed from the atmosphere but ~150 GtC is added. It should be reasonably obvious to see that a CO2 molecule spends, on average, ~5 years in the atmosphere. This is not the same as the time taken to remove an 'impulse' of CO2 from the atmosphere. I posted details of a very rough but very simple model on this blog some time back. Peter Dietze( a sceptic) has produced a more sophisticated model which is described in a paper on the John Daly site here www.john-daly.com/forcing/moderr.htm Dietze says Multiplication of T by ln(2) yields a half-life time of about 38 years. So any CO2 impulse injected into the atmosphere will take about 38 years to be reduced to half the original valueIf I remember rightly this is about the same as I got. Basically Dietze is saying that, if we stopped emitting CO2 from fossil fuel burning now, it will take ~38 years to remove 50 ppm of the 100 ppm that has been added since ~1850. The rate of decay does get slower, i.e. it is not a linear, so you cannot assume the 'excess' will be gone in 76 years for example. |
|
|
|
Post by dogsbody on May 19, 2010 11:21:26 GMT
Oh of course socold. Everyone is wrong except IPCC, and you. There is Solomon's 1000 year Reich nonsense too. Why was it again there is no necessity for the tropical troposphere "hot spot" despite all GCM's predicting it? Please, no barf mulch from ReinventedClimate. And why isn't the stratosphere cooling as advertised it should? Is there anything at all that is falsifiable?  You need to be careful here. I'd like to know eactly what those studies say. Socold's right there is a lot of confusion about this issue. There is a big difference between the turnover time and the time taken to remove 'excess' CO2 from the atmosphere. The turnover time or average life time of a CO2 molecule is ~5 years. This is easy enough to demonstrate. The atmosphere holds about 750 GtC (gigatons of carbon). During the annual cycle ~150 GtC is removed from the atmosphere but ~150 GtC is added. It should be reasonably obvious to see that a CO2 molecule spends, on average, ~5 years in the atmosphere. This is not the same as the time taken to remove an 'impulse' of CO2 from the atmosphere. I posted details of a very rough but very simple model on this blog some time back. Peter Dietze( a sceptic) has produced a more sophisticated model which is described in a paper on the John Daly site here www.john-daly.com/forcing/moderr.htm Dietze says Multiplication of T by ln(2) yields a half-life time of about 38 years. So any CO2 impulse injected into the atmosphere will take about 38 years to be reduced to half the original valueIf I remember rightly this is about the same as I got. Basically Dietze is saying that, if we stopped emitting CO2 from fossil fuel burning now, it will take ~38 years to remove 50 ppm of the 100 ppm that has been added since ~1850. The rate of decay does get slower, i.e. it is not a linear, so you cannot assume the 'excess' will be gone in 76 years for example. While your extrapolation is quite reasonable it depends on your original assumption being correct. The graph from Magellan was not CO2Science's graph, it is Segalstads. You may find some clarification by reading this Abstract from Essingh's paper. Attachments:
|
|
|
|
Post by socold on May 19, 2010 21:46:51 GMT
Unsurprisingly this claim was helpfully forwarded by skeptics to the EPA who were then able to get experts, at taxpayer expense, to spend time writing a detailed response. As I suspected the skeptics are confusing two separate concepts.
With regard to the part I have highlighted in red, judge for yourselves the plausibility that Segalstad is unaware of all this stuff - he certainly doesn't mention it in his attack on the IPCC.
EPA Reply To Comments - Comment (2-3)
Several commenters state that CO2 has a short lifetime in the atmosphere (0711.1, 0714.1): for example, a commenter (1616) claims that the lifetime of CO2 can be at most 20 years based on the 12% annual exchange of CO2 with the surface ocean and 10% exchange between the surface and deep ocean as shown in the National Aeronautics and Space Administration (NASA) carbon cycle diagram, and two commenters (3440.1, 3722) state that the overwhelming majority of scientific papers support a residence time of seven years in contrast to the TSD and IPCC. Several commenters (e.g. 3722) cite Professor Segalstad who has stated, based on his work on CO2 residence times (Segalstad 1997), that the assumption of a 50- to 200-year lifetime by IPCC results in a “missing sink” of 3 Gt of carbon a year, which is evidence that IPCC is mistaken. Another commenter submitted Essenhigh (2009), which developed a box model and also found that the lifetime of CO2 was on the order of a few years.
EPA Reply To Comments - Response (2-3)
EPA reviewed the information presented, as well as the work by Segalstad, and finds that it does not address the lifetime of a change in atmospheric concentration of CO2, but rather the lifetime in the atmosphere of an individual molecule of CO2. These are two different concepts. As stated in the First IPCC Scientific Assessment, “The turnover time of CO2 in the atmosphere, measured as the ratio of the content to the fluxes through it, is about 4 years. This means that on average it takes only a few years before a CO2 molecule in the atmosphere is taken up by plants or dissolved in the ocean. This short time scale must not be confused with the time it takes for the atmospheric CO2 level to adjust to a new equilibrium if sources or sinks change. This adjustment time ... is of the order of 50–200 years, determined mainly by the slow exchange of carbon between surface waters and the deep ocean” (Watson et al., 1990). The magnitudes of these large balanced sources and sinks are addressed in response 2-2, and are similar to those represented in the NASA carbon cycle diagram. Newer research has only extended and confirmed this statement from the first IPCC assessment report (Denman et al., 2007). A recent approximation for this perturbation lifetime is sometimes represented as the sum of decay functions with timescales of 1.9 years for a quarter of the CO2 emissions, 18.5 years for a third of the CO2, 173 years for a fifth of the CO2, and a constant term representing a nearly permanent increase for the remaining fifth (Forster et al., 2007).
The “missing sink” that was referred to by a commenter is also addressed in response 2-2, and is now called the “residual land sink.” The magnitude of this sink is about 2.6 Gt of carbon per year, with significant uncertainty. Denman et al. (2007) included a hypothesis that a portion of this sink is due to the increased growth of undisturbed tropical forest due to CO2 fertilization, but the carbon accumulation of natural systems is hard to quantify directly. The uncertainty in determining the size and nature of this residual sink does not contradict the assessment literature conclusions about the perturbation lifetime of CO2 concentration changes in the atmosphere, but is reflected in the carbon cycle uncertainty for future projections of CO2 (see responses regarding carbon cycle uncertainty in Volume 4 on future projections). The box model in Essenhigh (2009) is clearly flawed: the results from this model as reported in the paper include a lifetime for CO2 containing the 14C isotope that is a factor of 3 different from the lifetime of CO2 containing the 12C isotope. This difference in lifetimes is not scientifically compatible with the immense difficulty involved in isotope separation. The model assumes that each “control volume” (each volume represents either the ecosystem, the surface ocean, or the deep ocean) is perfectly mixed, which is contrary to the observations of oceanic CO2 which show that storage of carbon in the ocean is only at 15% of the equilibrium value, and that the mixing time between the surface ocean and intermediate and deep oceans is on the order of years to centuries (Field and Raupach, 2004). Additionally, the paper uses only historical fossil fuel emissions of CO2, without including land use change CO2, and contains the same confusion about “residence lifetime” and “adjustment lifetime” that has been addressed above. A common analogy used for CO2 concentrations is water in a bathtub. If the drain and the spigot are both large and perfectly balanced, then the time than any individual water molecule spends in the bathtub is short. But if a cup of water is added to the bathtub, the change in volume in the bathtub will persist even when all the water molecules originally from that cup have flowed out the drain. This is not a perfect analogy: in the case of CO2, there are several linked bathtubs, and the increased pressure of water in one bathtub from an extra cup will actually lead to a small increase in flow through the drain, so eventually the cup of water will be spread throughout the bathtubs leading to a small increase in each, but the point remains that the "residence time" of a molecule of water will be very different from the "adjustment time" of the bathtub as a whole. This analogy does not hold for other GHGs: methane, HFCs, and N2O are actually destroyed chemically in the atmosphere, unlike CO2 where the carbon is not destroyed but merely shifted from one reservoir to another, and therefore the residence lifetime of these gases is fairly close to the adjustment lifetime of their concentrations in the atmosphere. Similarly, any given molecule of CO2 is only expected to stay in the atmosphere for a few years before it moves into the oceans or ecosystem, but the change in atmospheric concentration due to combustion of fossil fuels can persist for much longer. Indeed, because the oceans and ecosystems are finite, some small fraction of CO2 emissions will have a perturbation lifetime in the atmosphere of thousands of years (Karlet al., 2009).
|
|
|
|
Post by magellan on May 19, 2010 22:47:47 GMT
Unsurprisingly this claim was helpfully forwarded by skeptics to the EPA who were then able to get experts, at taxpayer expense, to spend time writing a detailed response. As I suspected the skeptics are confusing two separate concepts. With regard to the part I have highlighted in red, judge for yourselves the plausibility that Segalstad is unaware of all this stuff - he certainly doesn't mention it in his attack on the IPCC. EPA Reply To Comments - Comment (2-3)Several commenters state that CO2 has a short lifetime in the atmosphere (0711.1, 0714.1): for example, a commenter (1616) claims that the lifetime of CO2 can be at most 20 years based on the 12% annual exchange of CO2 with the surface ocean and 10% exchange between the surface and deep ocean as shown in the National Aeronautics and Space Administration (NASA) carbon cycle diagram, and two commenters (3440.1, 3722) state that the overwhelming majority of scientific papers support a residence time of seven years in contrast to the TSD and IPCC. Several commenters (e.g. 3722) cite Professor Segalstad who has stated, based on his work on CO2 residence times (Segalstad 1997), that the assumption of a 50- to 200-year lifetime by IPCC results in a “missing sink” of 3 Gt of carbon a year, which is evidence that IPCC is mistaken. Another commenter submitted Essenhigh (2009), which developed a box model and also found that the lifetime of CO2 was on the order of a few years. EPA Reply To Comments - Response (2-3)EPA reviewed the information presented, as well as the work by Segalstad, and finds that it does not address the lifetime of a change in atmospheric concentration of CO2, but rather the lifetime in the atmosphere of an individual molecule of CO2. These are two different concepts. As stated in the First IPCC Scientific Assessment, “The turnover time of CO2 in the atmosphere, measured as the ratio of the content to the fluxes through it, is about 4 years. This means that on average it takes only a few years before a CO2 molecule in the atmosphere is taken up by plants or dissolved in the ocean. This short time scale must not be confused with the time it takes for the atmospheric CO2 level to adjust to a new equilibrium if sources or sinks change. This adjustment time ... is of the order of 50–200 years, determined mainly by the slow exchange of carbon between surface waters and the deep ocean” (Watson et al., 1990). The magnitudes of these large balanced sources and sinks are addressed in response 2-2, and are similar to those represented in the NASA carbon cycle diagram. Newer research has only extended and confirmed this statement from the first IPCC assessment report (Denman et al., 2007). A recent approximation for this perturbation lifetime is sometimes represented as the sum of decay functions with timescales of 1.9 years for a quarter of the CO2 emissions, 18.5 years for a third of the CO2, 173 years for a fifth of the CO2, and a constant term representing a nearly permanent increase for the remaining fifth (Forster et al., 2007). The “missing sink” that was referred to by a commenter is also addressed in response 2-2, and is now called the “residual land sink.” The magnitude of this sink is about 2.6 Gt of carbon per year, with significant uncertainty. Denman et al. (2007) included a hypothesis that a portion of this sink is due to the increased growth of undisturbed tropical forest due to CO2 fertilization, but the carbon accumulation of natural systems is hard to quantify directly. The uncertainty in determining the size and nature of this residual sink does not contradict the assessment literature conclusions about the perturbation lifetime of CO2 concentration changes in the atmosphere, but is reflected in the carbon cycle uncertainty for future projections of CO2 (see responses regarding carbon cycle uncertainty in Volume 4 on future projections). The box model in Essenhigh (2009) is clearly flawed: the results from this model as reported in the paper include a lifetime for CO2 containing the 14C isotope that is a factor of 3 different from the lifetime of CO2 containing the 12C isotope. This difference in lifetimes is not scientifically compatible with the immense difficulty involved in isotope separation. The model assumes that each “control volume” (each volume represents either the ecosystem, the surface ocean, or the deep ocean) is perfectly mixed, which is contrary to the observations of oceanic CO2 which show that storage of carbon in the ocean is only at 15% of the equilibrium value, and that the mixing time between the surface ocean and intermediate and deep oceans is on the order of years to centuries (Field and Raupach, 2004). Additionally, the paper uses only historical fossil fuel emissions of CO2, without including land use change CO2, and contains the same confusion about “residence lifetime” and “adjustment lifetime” that has been addressed above. A common analogy used for CO2 concentrations is water in a bathtub. If the drain and the spigot are both large and perfectly balanced, then the time than any individual water molecule spends in the bathtub is short. But if a cup of water is added to the bathtub, the change in volume in the bathtub will persist even when all the water molecules originally from that cup have flowed out the drain. This is not a perfect analogy: in the case of CO2, there are several linked bathtubs, and the increased pressure of water in one bathtub from an extra cup will actually lead to a small increase in flow through the drain, so eventually the cup of water will be spread throughout the bathtubs leading to a small increase in each, but the point remains that the "residence time" of a molecule of water will be very different from the "adjustment time" of the bathtub as a whole. This analogy does not hold for other GHGs: methane, HFCs, and N2O are actually destroyed chemically in the atmosphere, unlike CO2 where the carbon is not destroyed but merely shifted from one reservoir to another, and therefore the residence lifetime of these gases is fairly close to the adjustment lifetime of their concentrations in the atmosphere. Similarly, any given molecule of CO2 is only expected to stay in the atmosphere for a few years before it moves into the oceans or ecosystem, but the change in atmospheric concentration due to combustion of fossil fuels can persist for much longer. Indeed, because the oceans and ecosystems are finite, some small fraction of CO2 emissions will have a perturbation lifetime in the atmosphere of thousands of years (Karlet al., 2009). Are you ever going to cite your references with links? You do realize of course the "missing sink" in the second section still invalidates climate model predictions, and is not verifiable. Nothing you've posted invalidates Segalstad's. It's much like the IPCC rewriting history by eliminating the MWP and LIA by simply making them disappear based on a fraud fudge (for the politically correct). pubs.acs.org/doi/abs/10.1021/ef800581rThe driver for this study is the wide-ranging published values of the CO2 atmospheric residence time (RT), τ, with the values differing by more than an order of magnitude, where the significance of the difference relates to decisions on whether (1) to attempt control of combustion-sourced (anthropogenic) CO2 emissions, if τ > 100 years, or (2) not to attempt control, if τ 10 years. This given difference is particularly evident in the IPCC First 1990 Climate Change Report where, in the opening policymakers summary of the report, the RT is stated to be in the range of 50−200 years, and (largely) on the basis of that, it was also concluded in the report and from subsequent related studies that the current rising level of CO2 was due to combustion of fossil fuels, thus carrying the, now widely accepted, rider that CO2 emissions from combustion should therefore be curbed. However, the actual data in the text of the IPCC report separately states a value of 4 years. The differential of these two times is then clearly identified in the relevant supporting documents of the report as being, separately (1) a long-term (100 years) adjustment or response time to accommodate imbalance increases in CO2 emissions from all sources and (2) the actual RT in the atmosphere of 4 years. As a check on that differentiation and its alternative outcome, the definition and determination of RT thus defined the need for and focus of this study. In this study, using the combustion/chemical-engineering perfectly stirred reactor (PSR) mixing structure or 0D box for the model basis, as an alternative to the more commonly used global circulation models (GCMs), to define and determine the RT in the atmosphere and then using data from the IPCC and other sources for model validation and numerical determination, the data (1) support the validity of the PSR model application in this context and, (2) from the analysis, provide (quasi-equilibrium) RTs for CO2 of 5 years carrying C12 and 16 years carrying C14, with both values essentially in agreement with the IPCC short-term (4 year) value and, separately, in agreement with most other data sources, notably, a 1998 listing by Segalstad of 36 other published values, also in the range of 5−15 years. Additionally, the analytical results also then support the IPCC analysis and data on the longer “adjustment time” (100 years) governing the long-term rising “quasi-equilibrium” concentration of CO2 in the atmosphere. For principal verification of the adopted PSR model, the data source used was the outcome of the injection of excess 14CO2 into the atmosphere during the A-bomb tests in the 1950s/1960s, which generated an initial increase of approximately 1000% above the normal value and which then declined substantially exponentially with time, with τ = 16 years, in accordance with the (unsteady-state) prediction from and jointly providing validation for the PSR analysis. With the short (5−15 year) RT results shown to be in quasi-equilibrium, this then supports the (independently based) conclusion that the long-term (100 year) rising atmospheric CO2 concentration is not from anthropogenic sources but, in accordance with conclusions from other studies, is most likely the outcome of the rising atmospheric temperature, which is due to other natural factors. This further supports the conclusion that global warming is not anthropogenically driven as an outcome of combustion. The economic and political significance of that conclusion will be self-evident. |
|
|
|
Post by glc on May 20, 2010 9:16:17 GMT
Magellan: I've read your link, i.e. pubs.acs.org/doi/abs/10.1021/ef800581rfrom wghich you've highlighted this bit. However, the actual data in the text of the IPCC report separately states a value of 4 years......Socold's link answers this here As stated in the First IPCC Scientific Assessment, “The turnover time of CO2 in the atmosphere, measured as the ratio of the content to the fluxes through it, is about 4 yearsThe "4 years" clearly relates to the turnover time This is not the same as the time taken to remove the excess due to an impulse of CO2, i.e. the residence time (I think they call it). I referred to Dietze in an earlier post. He reckons about 50% will be remaining after 38 years and the e-folding time is 55 years. The e-folding time is where a proportion of 1/e (~37%) remains. The IPCC figures of between 50 and 200 years are probably not that far out. What you've got to remember is that, although some of the impulse excess from fossil fuel burning may still be present in 100 years, it will be well below "problem levels" - and will have been so for several decades. If Dietze's figures are close to being right and we stop burning fossil fuels to-day in 2010, the atmospheric CO2 level would be down to around 320-330 ppm by 2060. There is absoutely no chance that CO2 concentrations would be down to pre-industrial levels within 4 or 5 years. That is a huge amount of CO2 to be absorbed. Re: the missinng sink. There is one, and although no-one is quite sure what it is, it seems to behave quite predictably. Basically the earth seems to partly offset additional human emissions. If we emit the equivalent of 4ppm of CO2 the biosphere takes up an extra ~2ppm (note - this is not exact and varies slightly due to ENSO etc). The point is that this can be incorporated into any model without too much fuss so most predictions account for the 'sink'. Perhaps some of the early (e.g. 570 years) predictions didn't. |
|
|
|
Post by dogsbody on May 20, 2010 9:41:26 GMT
Unsurprisingly this claim was helpfully forwarded by skeptics to the EPA who were then able to get experts, at taxpayer expense, to spend time writing a detailed response. As I suspected the skeptics are confusing two separate concepts. With regard to the part I have highlighted in red, judge for yourselves the plausibility that Segalstad is unaware of all this stuff - he certainly doesn't mention it in his attack on the IPCC. EPA Reply To Comments - Comment (2-3)Several commenters state that CO2 has a short lifetime in the atmosphere (0711.1, 0714.1): for example, a commenter (1616) claims that the lifetime of CO2 can be at most 20 years based on the 12% annual exchange of CO2 with the surface ocean and 10% exchange between the surface and deep ocean as shown in the National Aeronautics and Space Administration (NASA) carbon cycle diagram, and two commenters (3440.1, 3722) state that the overwhelming majority of scientific papers support a residence time of seven years in contrast to the TSD and IPCC. Several commenters (e.g. 3722) cite Professor Segalstad who has stated, based on his work on CO2 residence times (Segalstad 1997), that the assumption of a 50- to 200-year lifetime by IPCC results in a “missing sink” of 3 Gt of carbon a year, which is evidence that IPCC is mistaken. Another commenter submitted Essenhigh (2009), which developed a box model and also found that the lifetime of CO2 was on the order of a few years. EPA Reply To Comments - Response (2-3)EPA reviewed the information presented, as well as the work by Segalstad, and finds that it does not address the lifetime of a change in atmospheric concentration of CO2, but rather the lifetime in the atmosphere of an individual molecule of CO2. These are two different concepts. As stated in the First IPCC Scientific Assessment, “The turnover time of CO2 in the atmosphere, measured as the ratio of the content to the fluxes through it, is about 4 years. This means that on average it takes only a few years before a CO2 molecule in the atmosphere is taken up by plants or dissolved in the ocean. This short time scale must not be confused with the time it takes for the atmospheric CO2 level to adjust to a new equilibrium if sources or sinks change. This adjustment time ... is of the order of 50–200 years, determined mainly by the slow exchange of carbon between surface waters and the deep ocean” (Watson et al., 1990). The magnitudes of these large balanced sources and sinks are addressed in response 2-2, and are similar to those represented in the NASA carbon cycle diagram. Newer research has only extended and confirmed this statement from the first IPCC assessment report (Denman et al., 2007). A recent approximation for this perturbation lifetime is sometimes represented as the sum of decay functions with timescales of 1.9 years for a quarter of the CO2 emissions, 18.5 years for a third of the CO2, 173 years for a fifth of the CO2, and a constant term representing a nearly permanent increase for the remaining fifth (Forster et al., 2007). The “missing sink” that was referred to by a commenter is also addressed in response 2-2, and is now called the “residual land sink.” The magnitude of this sink is about 2.6 Gt of carbon per year, with significant uncertainty. Denman et al. (2007) included a hypothesis that a portion of this sink is due to the increased growth of undisturbed tropical forest due to CO2 fertilization, but the carbon accumulation of natural systems is hard to quantify directly. The uncertainty in determining the size and nature of this residual sink does not contradict the assessment literature conclusions about the perturbation lifetime of CO2 concentration changes in the atmosphere, but is reflected in the carbon cycle uncertainty for future projections of CO2 (see responses regarding carbon cycle uncertainty in Volume 4 on future projections). The box model in Essenhigh (2009) is clearly flawed: the results from this model as reported in the paper include a lifetime for CO2 containing the 14C isotope that is a factor of 3 different from the lifetime of CO2 containing the 12C isotope. This difference in lifetimes is not scientifically compatible with the immense difficulty involved in isotope separation. The model assumes that each “control volume” (each volume represents either the ecosystem, the surface ocean, or the deep ocean) is perfectly mixed, which is contrary to the observations of oceanic CO2 which show that storage of carbon in the ocean is only at 15% of the equilibrium value, and that the mixing time between the surface ocean and intermediate and deep oceans is on the order of years to centuries (Field and Raupach, 2004). Additionally, the paper uses only historical fossil fuel emissions of CO2, without including land use change CO2, and contains the same confusion about “residence lifetime” and “adjustment lifetime” that has been addressed above. A common analogy used for CO2 concentrations is water in a bathtub. If the drain and the spigot are both large and perfectly balanced, then the time than any individual water molecule spends in the bathtub is short. But if a cup of water is added to the bathtub, the change in volume in the bathtub will persist even when all the water molecules originally from that cup have flowed out the drain. This is not a perfect analogy: in the case of CO2, there are several linked bathtubs, and the increased pressure of water in one bathtub from an extra cup will actually lead to a small increase in flow through the drain, so eventually the cup of water will be spread throughout the bathtubs leading to a small increase in each, but the point remains that the "residence time" of a molecule of water will be very different from the "adjustment time" of the bathtub as a whole. This analogy does not hold for other GHGs: methane, HFCs, and N2O are actually destroyed chemically in the atmosphere, unlike CO2 where the carbon is not destroyed but merely shifted from one reservoir to another, and therefore the residence lifetime of these gases is fairly close to the adjustment lifetime of their concentrations in the atmosphere. Similarly, any given molecule of CO2 is only expected to stay in the atmosphere for a few years before it moves into the oceans or ecosystem, but the change in atmospheric concentration due to combustion of fossil fuels can persist for much longer. Indeed, because the oceans and ecosystems are finite, some small fraction of CO2 emissions will have a perturbation lifetime in the atmosphere of thousands of years (Karlet al., 2009). Essingh also deals with your (probably realclimates) and the IPcc comments and interpretation. I add some xtracts from Essinghs extract. Commenting on Solomon et al (IPCC) This given difference is particularly evident in the IPCC First 1990 Climate Change Report where, in the opening policymakers summary of the report, the RT is stated to be in the range of 50−200 years, and (largely) on the basis of that, it was also concluded in the report and from subsequent related studies that the current rising level of CO2 was due to combustion of fossil fuels, thus carrying the, now widely accepted, rider that CO2 emissions from combustion should therefore be curbed. However, the actual data in the text of the IPCC report separately states a value of 4 years. . In this study, using the combustion/chemical-engineering perfectly stirred reactor (PSR) mixing structure or 0D box for the model basis, as an alternative to the more commonly used global circulation models (GCMs), to define and determine the RT in the atmosphere and then using data from the IPCC and other sources for model validation and numerical determination, the data (1) support the validity of the PSR model application in this context and, (2) from the analysis, provide (quasi-equilibrium) RTs for CO2 of 5 years carrying C12 and 16 years carrying C14, with both values essentially in agreement with the IPCC short-term (4 year) value and, separately, in agreement with most other data sources, notably, a 1998 listing by Segalstad of 36 other published values, also in the range of 5−15 years. Additionally, the analytical results also then support the IPCC analysis and data on the longer “adjustment time” (100 years) governing the long-term rising “quasi-equilibrium” concentration of CO2 in the atmosphere. With the short (5−15 year) RT results shown to be in quasi-equilibrium, this then supports the (independently based) conclusion that the long-term (100 year) rising atmospheric CO2 concentration is not from anthropogenic sources but, in accordance with conclusions from other studies, is most likely the outcome of the rising atmospheric temperature, which is due to other natural factors. This further supports the conclusion that global warming is not anthropogenically driven as an outcome of combustion. The economic and political significance of that conclusion will be self-evident. |
|
|
|
Post by nautonnier on May 20, 2010 14:07:05 GMT
Dogsbody:
"With the short (5−15 year) RT results shown to be in quasi-equilibrium, this then supports the (independently based) conclusion that the long-term (100 year) rising atmospheric CO2 concentration is not from anthropogenic sources but, in accordance with conclusions from other studies, is most likely the outcome of the rising atmospheric temperature, which is due to other natural factors. This further supports the conclusion that global warming is not anthropogenically driven as an outcome of combustion. The economic and political significance of that conclusion will be self-evident."
I would think that the CO2 rise is solely due to the rise in OCEAN temperature. Then in line with Henry's law the solubility of CO2 reduces and it outgasses from the oceans. Its residence time in the atmosphere everyone agrees with at 4 - 7 years scrubbed out by rain.
So as ocean temperatures have risen since the little ice age one symptom of this is the atmospheric concentration of CO2 should increase. The time baseline for this seems to fit the time baseline used by AGW to claim the 'start of the rise' that is blamed on industrialization. This 'physical law' means that warming oceans leads to rising CO2 levels with a time lag. This matches with the geological records.
This does not explain the warming - but it explains the rise in atmospheric CO2.
|
|
|
|
Post by socold on May 20, 2010 19:28:04 GMT
EPA response to comment 2-2 addresses that. The response is very long (addresses all points plus a lot of reference to additional positive evidence), so I include only the part that addresses the relevant comment (marked in red) Comment (2-2):A large number of commenters expressed doubt about the anthropogenic origins of the recent increase in CO2. Some commenters believe that humans produce a very small fraction of carbon dioxide (CO2), and thus have not contributed to CO2 rise (0153, 0247, 0425, 0455, 0498, 5858, 7022). Some propose various fractions—2.4% (0153), <1% (0425), 0.117% (1016.1, 1216.1) or 3% (0247, 5858, 9798)—and some state that human CO2 emissions are outweighed by CO2 from the ocean (0425, 0759) or volcanoes (0368, 0455, 2992) or outgassing (8978, referencing Khilyuk and Chilingar, 2006, on CO2 emissions from the Earth’s mantle). One commenter (0339) notes that the modern correlation between ocean temperature and CO2 levels indicates that oceans may be the main cause of CO2 increases, as does a commenter (1616) who states that “it is equally plausible that increased CO2 is caused by warming, rather than the other way around” based on the work of Robert Essenhigh. One commenter (1924) cites Spencer on the carbon-13/carbon-12 (13C/12C) interannual dilution being the same as natural variability, and on anthropogenic CO2 emissions being twice the observed atmospheric increase, and asks how high atmospheric levels of CO2 are the clear result of anthropogenic emissions. Finally, a commenter (11454.1) provided a quote from Heaven and Earth (Plimer, 2009) claiming that “Volcanoes produce more CO2 than the world’s cars and industries combined.”"[/QUOTE] Response (2-2):... With respect to the specific issues raised by commenters: Ocean CO2 emissions, and ocean temperature and CO2 links. Solubility of CO2 in the oceans does decrease with increasing temperature; however, the historical record indicates that even large temperature changes such as the glacial-interglacial transition result in changes of less than 100 parts per million (ppm) of CO2, and according to Denman et al., “A 1°C increase in sea surface temperature produces an increase in pCO2 of 6.9 to 10.2 ppm after 100 to 1,000 years,” making it hard to explain how the temperature changes over the past few centuries could lead to a CO2 change of 110 ppm since the preindustrial era. In addition, the oceans are serving as a net sink of CO2, as demonstrated by increasing acidity, rather than a net source. Therefore, we do not find commenter’s statements, including the reference to Essenhigh, to be plausible or supported by the literature. ... www.epa.gov/climatechange/endangerment.html |
|
|
|
Post by dogsbody on May 21, 2010 8:29:01 GMT
Dogsbody: "With the short (5−15 year) RT results shown to be in quasi-equilibrium, this then supports the (independently based) conclusion that the long-term (100 year) rising atmospheric CO2 concentration is not from anthropogenic sources but, in accordance with conclusions from other studies, is most likely the outcome of the rising atmospheric temperature, which is due to other natural factors. This further supports the conclusion that global warming is not anthropogenically driven as an outcome of combustion. The economic and political significance of that conclusion will be self-evident."I would think that the CO 2 rise is solely due to the rise in OCEAN temperature. Then in line with Henry's law the solubility of CO 2 reduces and it outgasses from the oceans. Its residence time in the atmosphere everyone agrees with at 4 - 7 years scrubbed out by rain. So as ocean temperatures have risen since the little ice age one symptom of this is the atmospheric concentration of CO 2 should increase. The time baseline for this seems to fit the time baseline used by AGW to claim the 'start of the rise' that is blamed on industrialization. This 'physical law' means that warming oceans leads to rising CO 2 levels with a time lag. This matches with the geological records. This does not explain the warming - but it explains the rise in atmospheric CO 2. Nautonier, I pretty much agree with your comments. I think the warming is directly related to solar activity, the cosmic ray cloud effect as described by Svensmark and supported by many solar and astrophysicists. Have another look at the graph that I have posted. You can see the evidence of Henry's law as you suggest. In the Elnino years warmer years the rate of mean annual CO2 increase is greater, in the Lanina cooler years the opposite occurs. Not sure how the warmers will explain this one, but I wait with great expectation. I read quickly through Socolds reply but had read it before as it's more or less the official IPCC position. However Socolds final reference to acidification is totally unscientific for several reasons. Firstly the 0.1 ph point movement towards a neutral ph falls within the IPCC's own range of natural variability. Secondly it hasn't been demonstrated that the shift is the result of more dissolved CO2. Thirdly: Even if it was it hasn't been demonstrated that it is from anthropogenic sources. For Socolds benefit I'll insert the graph of CO2/Temp/ElNino/LaNina again. After all this is Maxi's thread on Temp driving CO2. Attachments:
|
|
|
|
Post by icefisher on May 21, 2010 14:47:15 GMT
You need to be careful here. I'd like to know eactly what those studies say. Socold's right there is a lot of confusion about this issue. There is a big difference between the turnover time and the time taken to remove 'excess' CO2 from the atmosphere. The turnover time or average life time of a CO2 molecule is ~5 years. This is easy enough to demonstrate. The atmosphere holds about 750 GtC (gigatons of carbon). During the annual cycle ~150 GtC is removed from the atmosphere but ~150 GtC is added. It should be reasonably obvious to see that a CO2 molecule spends, on average, ~5 years in the atmosphere. This is not the same as the time taken to remove an 'impulse' of CO2 from the atmosphere. I posted details of a very rough but very simple model on this blog some time back. Peter Dietze( a sceptic) has produced a more sophisticated model which is described in a paper on the John Daly site here www.john-daly.com/forcing/moderr.htm Dietze says Multiplication of T by ln(2) yields a half-life time of about 38 years. So any CO2 impulse injected into the atmosphere will take about 38 years to be reduced to half the original valueIf I remember rightly this is about the same as I got. Basically Dietze is saying that, if we stopped emitting CO2 from fossil fuel burning now, it will take ~38 years to remove 50 ppm of the 100 ppm that has been added since ~1850. The rate of decay does get slower, i.e. it is not a linear, so you cannot assume the 'excess' will be gone in 76 years for example. Producing models on this topic for which so little reliable observational evidence exists pretty much speaks to what the problem essentially revolves around. CO2 is increasing at a steady rate its going from a microscopic teensy amount in the atmosphere to a microscopic teensy bit more. Its like all the bad guys are gone so lets worry about little bugs. Bottom line here is the carbon cycle is heavily influenced by living things since its an essential life giving gas. How fast plankton take up CO2 and drop to the bottom of the ocean may well be the primary exchange mechanism of the past. One company experimented with the idea of spreading iron dust on ocean waters to spur plankton growth after noting the limiting nutrient for plankton growth in the ocean was iron. Claims are the experiment failed because instead of the plankton living its happy existence then dutifully falling to the ocean floor it instead spurred a food chain explosion. Plankton eaters not surprisingly located these new rich sources of plankton and prospered then plankton eater eaters showed up. Talking about a real environmentalist nightmare considering who sits at the top of that food chain!!
|
|
|
|
Post by nautonnier on May 21, 2010 15:46:23 GMT
EPA response to comment 2-2 addresses that. The response is very long (addresses all points plus a lot of reference to additional positive evidence), so I include only the part that addresses the relevant comment (marked in red) Comment (2-2):A large number of commenters expressed doubt about the anthropogenic origins of the recent increase in CO2. Some commenters believe that humans produce a very small fraction of carbon dioxide (CO2), and thus have not contributed to CO2 rise (0153, 0247, 0425, 0455, 0498, 5858, 7022). Some propose various fractions—2.4% (0153), <1% (0425), 0.117% (1016.1, 1216.1) or 3% (0247, 5858, 9798)—and some state that human CO2 emissions are outweighed by CO2 from the ocean (0425, 0759) or volcanoes (0368, 0455, 2992) or outgassing (8978, referencing Khilyuk and Chilingar, 2006, on CO2 emissions from the Earth’s mantle). One commenter (0339) notes that the modern correlation between ocean temperature and CO2 levels indicates that oceans may be the main cause of CO2 increases, as does a commenter (1616) who states that “it is equally plausible that increased CO2 is caused by warming, rather than the other way around” based on the work of Robert Essenhigh. One commenter (1924) cites Spencer on the carbon-13/carbon-12 (13C/12C) interannual dilution being the same as natural variability, and on anthropogenic CO2 emissions being twice the observed atmospheric increase, and asks how high atmospheric levels of CO2 are the clear result of anthropogenic emissions. Finally, a commenter (11454.1) provided a quote from Heaven and Earth (Plimer, 2009) claiming that “Volcanoes produce more CO2 than the world’s cars and industries combined.”" ... With respect to the specific issues raised by commenters: Ocean CO2 emissions, and ocean temperature and CO2 links. Solubility of CO2 in the oceans does decrease with increasing temperature; however, the historical record indicates that even large temperature changes such as the glacial-interglacial transition result in changes of less than 100 parts per million (ppm) of CO2, and according to Denman et al., “A 1°C increase in sea surface temperature produces an increase in pCO2 of 6.9 to 10.2 ppm after 100 to 1,000 years,” making it hard to explain how the temperature changes over the past few centuries could lead to a CO2 change of 110 ppm since the preindustrial era. In addition, the oceans are serving as a net sink of CO2, as demonstrated by increasing acidity, rather than a net source. Therefore, we do not find commenter’s statements, including the reference to Essenhigh, to be plausible or supported by the literature. ... www.epa.gov/climatechange/endangerment.html[/quote] "however, the historical record indicates that even large temperature changes such as the glacial-interglacial transition result in changes of less than 100 parts per million (ppm) of CO2, and according to Denman et al., “A 1°C increase in sea
surface temperature produces an increase in pCO2 of 6.9 to 10.2 ppm after 100 to 1,000 years,"Using the totally spurious bubbles in ice-core record an invalid proxy. CO 2 will dissolve in the water at the poles and the air concentrataion be lower (shows in the satellite imagery from NOAA) and what CO 2 there is in ice bubble diffuses into the ice. |
|
|
|
Post by socold on May 21, 2010 19:03:59 GMT
Have another look at the graph that I have posted. You can see the evidence of Henry's law as you suggest. In the Elnino years warmer years CO2 levels rise, in the Lanina cooler years CO2 levels fall. Not sure how the warmers will explain this one, but I wait with great expectation. It's that simple - in warm years co2 rises faster and in cool years it rises slower. Why is it rising at all in the longterm? human emissions. It was noted above that the oceans is a net absorber of co2 so outgassing from the ocean is not a candidate for the rise. The oceans over the past century have limited rather than contributed to the rise. What range are you referring to? AR4 notes that "The mean pH of surface waters ranges between 7.9 and 8.3 in the open ocean", but that isn't a range of change over time, that's a change over space. This has been demonstrated. As the IPCC points out: "The uptake of anthropogenic carbon by the ocean changes the chemical equilibrium of the ocean. Dissolved CO2 forms a weak acid.1 As CO2 increases, pH decreases, that is, the ocean becomes more acidic. Ocean pH can be computed from measurements of DIC and alkalinity. A decrease in surface pH of 0.1 over the global ocean was calculated from the estimated uptake of anthropogenic carbon between 1750 and 1994 (Sabine et al., 2004b; Raven et al., 2005), with the lowest decrease (0.06) in the tropics and subtropics, and the highest decrease (0.12) at high latitudes, consistent with the lower buffer capacity of the high latitudes compared to the low latitudes." The origin of the atmospheric co2 rise (which is the source of the ocean carbon uptake) has been shown to be anthropogenic, as in Response (2-2) above: "As stated in CCSP (2007) “The cause of the recent increase in atmospheric CO2 is confirmed beyond a reasonable doubt.” There are many ways in which scientists determine the emissions associated with particular sources and activities. These are explained in detail in Chapter 7 of the Intergovernmental Panel on Climate Change’s (IPCC’s) Fourth Assessment Report (Denman et al., 2007), and they include isotope signatures, oxygen depletion, north/south gradient, and partitioning of excess carbon into sinks. As stated in the Third Assessment Report (IPCC, 2001c), “Several additional lines of evidence confirm that the recent and continuing increase of atmospheric CO2 content is caused by anthropogenic CO2 emissions— most importantly fossil fuel burning. First, atmospheric O2 is declining at a rate comparable with fossil fuel emissions of CO2 (combustion consumes O2). Second, the characteristic isotopic signatures of fossil fuel (its lack of 14C, and depleted content of 13C) leave their mark in the atmosphere. Third, the increase in observed CO2 concentration has been faster in the northern hemisphere, where most fossil fuel burning occurs.” |
|
|
|
Post by socold on May 21, 2010 19:09:44 GMT
Using the totally spurious bubbles in ice-core record an invalid proxy. CO 2 will dissolve in the water at the poles and the air concentrataion be lower (shows in the satellite imagery from NOAA) and what CO 2 there is in ice bubble diffuses into the ice. A commenter (0740.1) states that ice core CO2 measurements are impacted by water contamination, and that there are no other methods of measuring historical CO2 (commenter 3722 also objects to ice core record manipulation). Several commenters (0339, 0714.1, 2210.5, 3722) have cited either Beck (2007) or Jaworowski to support a contention that CO2 was at high concentrations in the recent past immediately before the Mauna Loa record started, or during past interglacials (0655). Response (2-4)We disagree with the assertion by several commenters that estimates of historical CO2 concentrations are incorrect. According to IPCC (Jansen et al., 2007), “it is possible to derive time series of atmospheric trace gases and aerosols for the period from about 650 kyr [thousand years] to the present from air trapped in polar ice and from the ice itself.” This methodology has been “verified against recent (i.e., post-1950) measurements made by direct instrumental sampling.” Additionally, these measurements are consistent with various less accurate methods such as using the size of stomatal pores on tree leaves, boron isotope measurements in plankton buried under the ocean, or carbon isotope ratios in algae buried in the ocean floors, moss samples, and foraminefera carbonate shells. Therefore, there is extremely high confidence in the CO2 values determined from the ice core records, and we disagree that there is any evidence that water contamination or other manipulations reduce the confidence in the ice core estimates. The commenters cited a theory from Jaworowski that water contamination in the ice core record reduces its reliability, and that the IPCC CO2 historical estimates require shifting the ice core records an arbitrary number of years in order to make them line up with the instrumental record. The critiques of Jaworowski on the shift were addressed by Hans Oeschger (1995), who pointed out that the ice core record shift was done in accordance with theoretical estimates of the rate of diffusion in gases in firn, and that these theoretical estimates were confirmed by isotopic enrichment in line with theory. Güllük et al. (1998) also rebutted Jaworowski on contamination, stating that “Jaworowski et al. [1992, 1994] suggested that CO2 measurements may be subject to fractionation due to clathrate formation and destruction. The good agreement of our CO2 measurements with those made by LGGE using the milling extraction procedure makes this artefact unlikely.” Similarly, Raynaud et al. (1993) found that the objections by Jaworowski were unfounded, demonstrating that the changes in CO2 and methane (CH4) are similar for different interglacial periods, regardless of depth, and that ice cores from different locations give the same values regardless of different “brittle zone” conditions between the different locations. |
|
|
|
Post by glc on May 21, 2010 19:40:21 GMT
Nautonier, I pretty much agree with your comments. I think the warming is directly related to solar activity, the cosmic ray cloud effect as described by Svensmark and supported by many solar and astrophysicists.
Have another look at the graph that I have posted. You can see the evidence of Henry's law as you suggest. In the Elnino years warmer years CO2 levels rise, in the Lanina cooler years CO2 levels fall. Not sure how the warmers will explain this one, but I wait with great expectation.
There's nothing to explain. Can we get this sorted once and for all. I've answered it - socold's answered it and it still doesn't seem to have sunk in.
CO2 LEVELS RISE EVERY YEAR. THEY RISE IN EL NINO YEARS. THEY ALSO RISE IN LA NINA YEARS. THEY DO NOT FALL. The only difference being that they rise faster in El Nino years than in La Nina years.
The increases in atmospheric CO2 since ~1850 is almost entirely due to fossil fuel burning.
|
|
maxi
New Member

Posts: 6
|
Post by maxi on May 21, 2010 20:21:20 GMT
Dogsbody: "With the short (5−15 year) RT results shown to be in quasi-equilibrium, this then supports the (independently based) conclusion that the long-term (100 year) rising atmospheric CO2 concentration is not from anthropogenic sources but, in accordance with conclusions from other studies, is most likely the outcome of the rising atmospheric temperature, which is due to other natural factors. This further supports the conclusion that global warming is not anthropogenically driven as an outcome of combustion. The economic and political significance of that conclusion will be self-evident."I would think that the CO 2 rise is solely due to the rise in OCEAN temperature. Then in line with Henry's law the solubility of CO 2 reduces and it outgasses from the oceans. Its residence time in the atmosphere everyone agrees with at 4 - 7 years scrubbed out by rain. So as ocean temperatures have risen since the little ice age one symptom of this is the atmospheric concentration of CO 2 should increase. The time baseline for this seems to fit the time baseline used by AGW to claim the 'start of the rise' that is blamed on industrialization. This 'physical law' means that warming oceans leads to rising CO 2 levels with a time lag. This matches with the geological records. This does not explain the warming - but it explains the rise in atmospheric CO 2. Nautonier, I pretty much agree with your comments. I think the warming is directly related to solar activity, the cosmic ray cloud effect as described by Svensmark and supported by many solar and astrophysicists. Have another look at the graph that I have posted. You can see the evidence of Henry's law as you suggest. In the Elnino years warmer years CO2 levels rise, in the Lanina cooler years CO2 levels fall. Not sure how the warmers will explain this one, but I wait with great expectation. I read quickly through Socolds reply but had read it before as it's more or less the official IPCC position. However Socolds final reference to acidification is totally unscientific for several reasons. Firstly the 0.1 ph point movement towards a neutral ph falls within the IPCC's own range of natural variability. Secondly it hasn't been demonstrated that the shift is the result of more dissolved CO2. Thirdly: Even if it was it hasn't been demonstrated that it is from anthropogenic sources. For Socolds benefit I'll insert the graph of CO2/Temp/ElNino/LaNina again. After all this is Maxi's thread on Temp driving CO2. I agree, let us go back to what this thread originally was about. Your additional graph just underlines the complete match between temperature and CO2 growth rate. That there is a connection is not that surprising, but CO2’s nearly immediate respond is surprising I still wonder what the physical explanation may be. About the Svensmark effect. Manny has tried to falsify it but nobody has hitherto succeeded. CERN is for the time being running a project, to show if prof. Svensmarks theory of how clouds are formed is right. |
|



