|
|
Post by Andrew on Sept 24, 2015 10:00:23 GMT
" ... 20 or so extra degrees provided by the GHE." Andrew, what do you mean by "extra"? I mean that the heat rays known as IR raise the temperature of the earth by many many tens of degrees just by virtue of their direct warming ability upon the Earths surface. The GHE then provides an additional warming of a few tens of degrees (also using IR) |
|
|
|
Post by missouriboy on Sept 24, 2015 13:10:04 GMT
" ... 20 or so extra degrees provided by the GHE." Andrew, what do you mean by "extra"? I mean that the heat rays known as IR raise the temperature of the earth by many many tens of degrees just by virtue of their direct warming ability upon the Earths surface. The GHE then provides an additional warming of a few tens of degrees (also using IR) The high (7000 feet), dry (very low humidity) climate of the Arizona end of the Colorado Plateau is a fine place to see what the sun will and will not do for your body. Summer night temperatures here will range from 45-55 degrees F (8-13 C). Morning strolls convinced me that I'm a solar energy absorber ... the exact combination of visible and near infra-red, as well as direct and reflected, is unknown ... but when the sun drops behind a cloud, you immediately feel the difference as your body feels the ambient temperature of the local prevailing air mass. Thermal infra-red, at wavelengths well beyond the attached chart, is re-radiated from objects composing the landscape surface, but does little heating in that dry air mass as it zips on out to space. The CO2 that's there seems to be of the cold-natured variety. 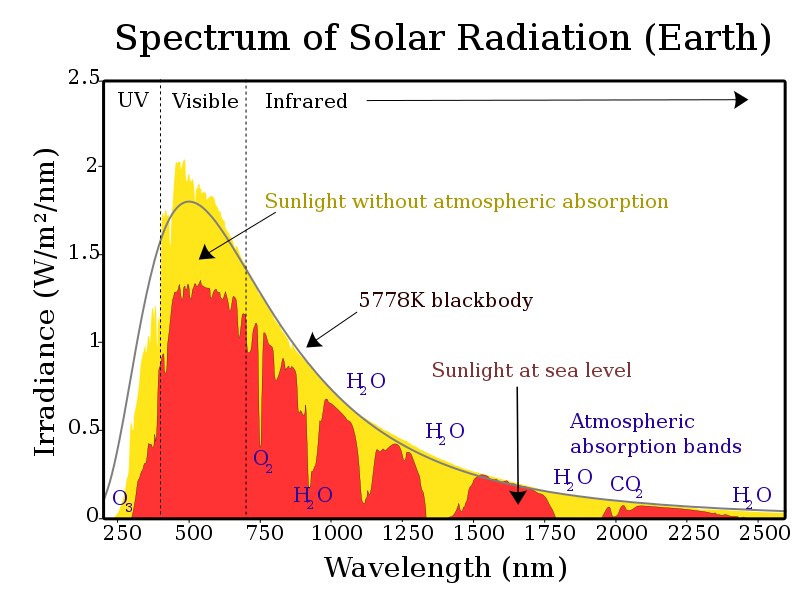 |
|
|
|
Post by sigurdur on Sept 24, 2015 15:01:38 GMT
You could replace all the H2o vapor with CO2 and you would either cook or freeze your ass off.
|
|
|
|
Post by acidohm on Sept 24, 2015 15:14:49 GMT
Tbh, when I initially found out the sun had cycles about 18 months ago, 2 of the next facts I discovered were tsi doesn't change much, uv does. That did it for me in that uv is what heats water, a very common molecule on this planet. Unlike co2, which effects ir, which can't heat anything. ... The variation of UV is not very large and mainly impacts the upper atmosphere which does get very hot but there is almost no atmosphere up there. IR is what you feel when you stand in front of any hot object. Ie heat rays that can cross distances even when you are not touching an object. The relative absence of IR is what you feel when you are cold. Without the heating effect of IR the planet would be extremely cold, and far far colder than the 20 or so extra degrees provided by the GHE. UV varies 10% between solar min and max (on average or whatever, obv depends on strength of max etc) IR penetrates 1mm of water and is done, UV penetrates many cm Ground gains and loses heat very easily, water gains and loses very slowly. |
|
|
|
Post by Andrew on Sept 24, 2015 16:44:47 GMT
The variation of UV is not very large and mainly impacts the upper atmosphere which does get very hot but there is almost no atmosphere up there. IR is what you feel when you stand in front of any hot object. Ie heat rays that can cross distances even when you are not touching an object. The relative absence of IR is what you feel when you are cold. Without the heating effect of IR the planet would be extremely cold, and far far colder than the 20 or so extra degrees provided by the GHE. UV varies 10% between solar min and max (on average or whatever, obv depends on strength of max etc) IR penetrates 1mm of water and is done, UV penetrates many cm Ground gains and loses heat very easily, water gains and loses very slowly. I think you are mixing up ideas there. Water is likely to gain heat very quickly but the surface layer of water cannot easily be heated or cooled quickly because it has access to/contact with the sub surface layers via IR and mixing. The solid surface does not have the access to distant layers via IR or mixing and is only able to transfer heat to and from the very next molecule of material via conduction |
|
|
|
Post by icefisher on Sept 24, 2015 18:03:45 GMT
" ... 20 or so extra degrees provided by the GHE." Andrew, what do you mean by "extra"? I mean that the heat rays known as IR raise the temperature of the earth by many many tens of degrees just by virtue of their direct warming ability upon the Earths surface. The GHE then provides an additional warming of a few tens of degrees (also using IR) IR is what you feel when you stand in front of a hot object? Are you claiming you cannot feel visible light? Heck Andrew I didn't know that lasers were impossible! Its hard to see what you are contributing to this discussion. |
|
|
|
Post by Andrew on Sept 24, 2015 18:44:30 GMT
I mean that the heat rays known as IR raise the temperature of the earth by many many tens of degrees just by virtue of their direct warming ability upon the Earths surface. The GHE then provides an additional warming of a few tens of degrees (also using IR) IR is what you feel when you stand in front of a hot object? Are you claiming you cannot feel visible light? Heck Andrew I didn't know that lasers were impossible! Its hard to see what you are contributing to this discussion. who pulled your chain? |
|
|
|
Post by sigurdur on Sept 24, 2015 18:53:43 GMT
UV varies 10% between solar min and max (on average or whatever, obv depends on strength of max etc) IR penetrates 1mm of water and is done, UV penetrates many cm Ground gains and loses heat very easily, water gains and loses very slowly. I think you are mixing up ideas there. Water is likely to gain heat very quickly but the surface layer of water cannot easily be heated or cooled quickly because it has access to/contact with the sub surface layers via IR and mixing. The solid surface does not have the access to distant layers via IR or mixing and is only able to transfer heat to and from the very next molecule of material via conduction Water, because of its physical properties gains heat at the same rate as a rock. Where it is different is HOW much heat per cubic inch it can gain, or conversely loose. |
|
|
|
Post by Andrew on Sept 24, 2015 18:55:28 GMT
I think you are mixing up ideas there. Water is likely to gain heat very quickly but the surface layer of water cannot easily be heated or cooled quickly because it has access to/contact with the sub surface layers via IR and mixing. The solid surface does not have the access to distant layers via IR or mixing and is only able to transfer heat to and from the very next molecule of material via conduction Water, because of its physical properties gains heat at the same rate as a rock. Where it is different is HOW much heat per cubic inch it can gain, or conversely loose. Good point. |
|
|
|
Post by acidohm on Sept 24, 2015 19:05:04 GMT
Apologies,,,i merely post this to clarify my previous statement which i just sort of blurted out, not trying to correct anyone posting previous to this... "The specific heat of water is 1 calorie/gram °C = 4.186 joule/gram °C which is higher than any other common substance. As a result, water plays a very important role in temperature regulation. The specific heat per gram for water is much higher than that for a metal for example" "The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius" Hence i said water gains and loses heat slowly....it's hard to heat it, and it then likes to hold on to the heat... As a plumber, soldering/brazing pipes is part of the job, the WORST thing is to have any water in the pipe, it absorbs all the heat, takes ages to evaporate and wicks all the heat from the metal...the solder will not melt on the metal for maybe minutes, as opposed to seconds when dry. Water absorbs alot of heat with little rise in temp. IR does NOT raise the temp of water...UV does. go in the sea on a hot day....what heated it?  why are you burning on the beach??? Plenty of UV at ground level. If most of the UV is absorbed in the upper atmosphere....ask, what effect does the absorption of all that energy do to the 95% of gases that effect global warming?? what happens if you have 50 years of even ,moderately above average solar activity or even just average..... Then take it away...... wattsupwiththat.com/2011/09/20/uv-low-during-recent-solar-minimum/I would rather find a more subjective source then WWUT which i read frequently...but, ya know, i like to appear impartial.... |
|
|
|
Post by sigurdur on Sept 24, 2015 19:35:52 GMT
Good job acid
|
|
|
|
Post by acidohm on Sept 24, 2015 19:58:35 GMT
Thx Sig....tbh I think I drank too much coffee today...I'm very excitable!
|
|
|
|
Post by Andrew on Sept 24, 2015 20:19:22 GMT
Apologies,,,i merely post this to clarify my previous statement which i just sort of blurted out, not trying to correct anyone posting previous to this... "The specific heat of water is 1 calorie/gram °C = 4.186 joule/gram °C which is higher than any other common substance. As a result, water plays a very important role in temperature regulation. The specific heat per gram for water is much higher than that for a metal for example" "The specific heat is the amount of heat per unit mass required to raise the temperature by one degree Celsius" Hence i said water gains and loses heat slowly....it's hard to heat it, and it then likes to hold on to the heat... As a plumber, soldering/brazing pipes is part of the job, the WORST thing is to have any water in the pipe, it absorbs all the heat, takes ages to evaporate and wicks all the heat from the metal...the solder will not melt on the metal for maybe minutes, as opposed to seconds when dry. Water absorbs alot of heat with little rise in temp. IR does NOT raise the temp of water...UV does. go in the sea on a hot day....what heated it?  why are you burning on the beach??? Plenty of UV at ground level. If most of the UV is absorbed in the upper atmosphere....ask, what effect does the absorption of all that energy do to the 95% of gases that effect global warming?? what happens if you have 50 years of even ,moderately above average solar activity or even just average..... Then take it away...... wattsupwiththat.com/2011/09/20/uv-low-during-recent-solar-minimum/I would rather find a more subjective source then WWUT which i read frequently...but, ya know, i like to appear impartial.... I am not sure what you are getting at here. Are you really saying that IR cannot raise the temperature of water?? |
|
|
|
Post by acidohm on Sept 24, 2015 20:32:16 GMT
Not more then the 1st mm at a time no
Well....Having said that ir is the largest spectrum.....educate me...
|
|
|
|
Post by icefisher on Sept 24, 2015 21:04:08 GMT
UV varies 10% between solar min and max (on average or whatever, obv depends on strength of max etc) IR penetrates 1mm of water and is done, UV penetrates many cm Ground gains and loses heat very easily, water gains and loses very slowly. I think you are mixing up ideas there. Water is likely to gain heat very quickly but the surface layer of water cannot easily be heated or cooled quickly because it has access to/contact with the sub surface layers via IR and mixing. The solid surface does not have the access to distant layers via IR or mixing and is only able to transfer heat to and from the very next molecule of material via conduction Andrew it might be useful to point out that all you are doing is disputing the language that Acidohm is using. It can be confusing to say that water gains heat very slowly and ground easily. However, it doesn't seem a stretch to understand he is talking about degrees temperature rather joules of heat. Heat and temperature are often not used precisely and often interchanged so you may as well be complaining he did not put a period at the end of a sentence. I read the sentence and was thinking temperature instead of joules also. It took a while to figure out what new infinitesimal point you were trying to make. It seems a more intelligent and imaginative individual would ask a question first, like "don't you mean temperature instead of heat?" if one was actually thinking he meant joules instead of degrees. I should point out at this point you made a boner too. Subsurface layers of water are affected not by IR and mixing, but by instead uniquely via UV, Visible Light, and mixing and indeed the solid surface does not have access to sublayers by anything except conduction. Probably the biggest piece of scientific BS passed around by the warmists is that lightwaves absorbed by the atmosphere have no impact on surface climate, except of course to the degree they need it to. Such deception regarding the state of science is central to the claim that variations in the sun does not in any significant part affect surface climate. But that seems disputed by the affects of solar cycles on surface warming rates. 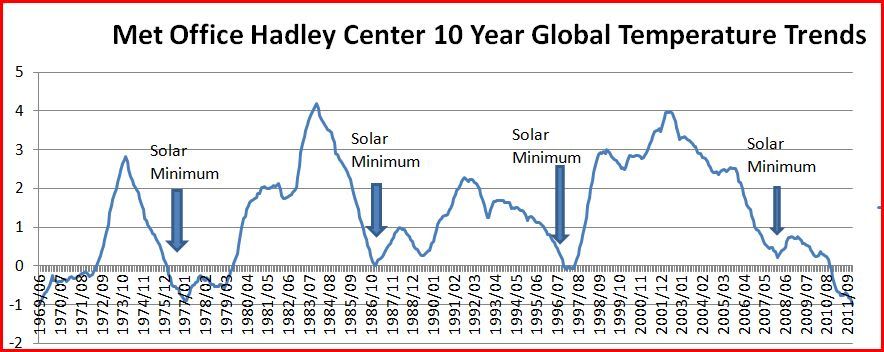 If somebody wants to actually dispute how a shifting of the solar light spectrum may or may not affect climate one needs to first explain the above chart. Poohing poohing solar variation has been a favorite pastime of warmists. First it had no measurable effect despite it being a NOAA favorite theory before Al Gore started firing people for believing in it. Then when the pause came the effect was selectively used as an explanation for the pause but not the initial warming. I am still trying to figure how Foster and Rahmstorf can turn on and off the effect whenever they choose. What I see in the above chart is a complete elimination of any warming effect by what ever was inducing it each and every solar cycle. This is using Phil Jones temperature data so its certainly not a skeptic invention. Why Phil Jones can't see the problem in his own data might be associated with either the amount of green in his pocket or grey matter in his skull. I am completely aware that the above chart which only depicts variation of surface warming is insufficient to explain the general warming over the past 100 years. That is where glacial and ocean rates of change and their equilibrium points help complicate the entire situation beyond any modeled theory to an extent that climate models have been unable to accurate hindcast ever, and currently can't forecast either. It seems clear to me that TSI variation has been discounted by you and the warmist community but I do not see how that is consistent with the above chart. If solar cycles lasted 40 years instead of 11 we would have opportunity to see their full effect on the surface and upper oceans which are the only elements currently in climate models. They won't include the solar cycles except when they try to explain the pause and even then its put in as an unquantified value so as to try to avoid an overall quantification in some smoke and mirrors study like the trash that Foster and Rahmstorf study whose basis has evaporated with the coming of an El Nino and Solar Maximum never to see the light of day again. The effects on this variation on the mid ocean, deep ocean, and glacial ice are much longer processes extending for thousands of years so in fact we don't know what the full effect of solar cycles are much less what effects longer periods of solar quietness is. I can't see a basis for handwaving anything away until we have a better understanding of natural variation. The theory that natural variation had been overridden, sold to us in IPCC's AR3, has been falsified and it rested on the concept of no natural variation currently occurring, yet the chart above proves they were lying. It was bolstered by the notion of omniscience (yes warmist scientists think they are Gods!) being sufficient to push political change. |
|