Understanding Water, Yes, it is quite complex
Sept 16, 2016 9:44:17 GMT
sigurdur and acidohm like this
Post by Ufasuperstorm on Sept 16, 2016 9:44:17 GMT
Water makes up .05 percent of the earths entire mass. If recent studies are accurate, the number may need to be revised up, as there are now hypothesized oceans worth of water in the mantle. Having so much of the water on the surface, means water is a moderator of our climate. It can be found in abundance in multiple states here on earth. I want to discuss some unique chemical/physical properties of water.
Let's start with the enthalpy of fusion. If you have 1 gram of water at 50 degrees Celsius and 1 gram of ice at 50 degrees Celsius you produce 2 grams of water at 0 degrees Celsius. It takes a lot of energy to break bonds on an atomic level and overcome ambient pressure in response to a change in volume. This is why water has an incredibly high enthalpy of fusion.
en.wikipedia.org/wiki/Enthalpy_of_fusion
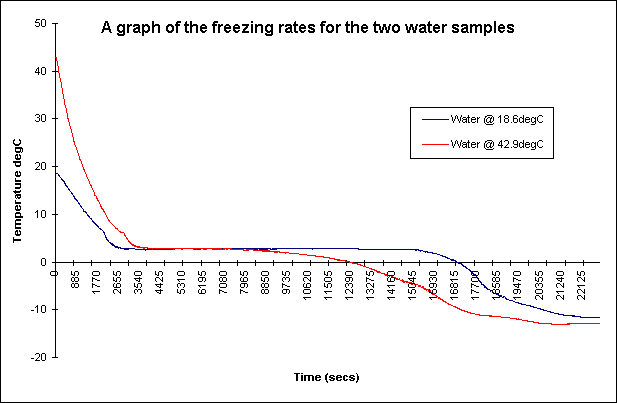
Since, we now brought up the enthalpy of fusion we must now discuss the Mpemba effect. Warm/hot water actually freezes faster than colder water at certain conditions. This effect has been well know since Aristotle's time but even today, I have seen various papers claiming a different central mechanism for this effect.
arxiv.org/ftp/arxiv/papers/1003/1003.3185.pdf
In retrospect, when water absorbs heat, covalent molecules get smaller and release there energy. This energy release is equal to cooling. You could say water has a built in temperature balancing mechanism?
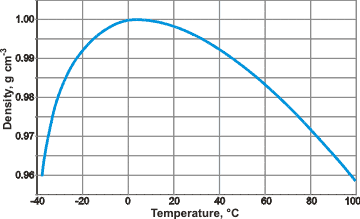
Furthermore, I would also like to point out the density of supercooled water at -38 degrees Celsius is the same as 98 degrees Celsius. Additionally, as supercooled water reaches 228 Kelvin it approaches the density of ice.
When you look on the atomic level physically/chemically what has to happen for water to possess these properties it is actually quite complex and not fully understood.
Water in various states exhibits a "retrograde behavior" and is indeed quite complex and worth discussing.
Let's start with the enthalpy of fusion. If you have 1 gram of water at 50 degrees Celsius and 1 gram of ice at 50 degrees Celsius you produce 2 grams of water at 0 degrees Celsius. It takes a lot of energy to break bonds on an atomic level and overcome ambient pressure in response to a change in volume. This is why water has an incredibly high enthalpy of fusion.
1) To heat 1 kg (1.00 liter) of water from 283.15 K to 303.15 K (10 °C to 30 °C) requires 83.6 kJ. However, to melt ice also requires energy. To heat 1kg of ice from 273.15 K to water at 293.15 K (0 °C to 20 °C) requires:
(1) 333.55 J/g (heat of fusion of ice) = 333.55 kJ/kg = 333.55 kJ for 1 kg of ice to melt
PLUS
(2) 4.18 J/(g·K) × 20K = 4.18 kJ/(kg·K) × 20K = 83.6 kJ for 1 kg of water to increase in temperature by 20 K
= 417.15 kJ
Thus one part ice at 0 °C will cool almost exactly 4 parts water from 20 °C to 0 °C.
(1) 333.55 J/g (heat of fusion of ice) = 333.55 kJ/kg = 333.55 kJ for 1 kg of ice to melt
PLUS
(2) 4.18 J/(g·K) × 20K = 4.18 kJ/(kg·K) × 20K = 83.6 kJ for 1 kg of water to increase in temperature by 20 K
= 417.15 kJ
Thus one part ice at 0 °C will cool almost exactly 4 parts water from 20 °C to 0 °C.
en.wikipedia.org/wiki/Enthalpy_of_fusion
Since, we now brought up the enthalpy of fusion we must now discuss the Mpemba effect. Warm/hot water actually freezes faster than colder water at certain conditions. This effect has been well know since Aristotle's time but even today, I have seen various papers claiming a different central mechanism for this effect.

We observed hot water freezing before cold water 28 times in 28 attempts under the conditions
described here.
described here.
arxiv.org/ftp/arxiv/papers/1003/1003.3185.pdf
In retrospect, when water absorbs heat, covalent molecules get smaller and release there energy. This energy release is equal to cooling. You could say water has a built in temperature balancing mechanism?
Furthermore, I would also like to point out the density of supercooled water at -38 degrees Celsius is the same as 98 degrees Celsius. Additionally, as supercooled water reaches 228 Kelvin it approaches the density of ice.

When you look on the atomic level physically/chemically what has to happen for water to possess these properties it is actually quite complex and not fully understood.
Water in various states exhibits a "retrograde behavior" and is indeed quite complex and worth discussing.

