|
|
Post by steve on Oct 15, 2009 16:17:26 GMT
No! And this is a key point. Noting that "funnel" was not a term I used, the "funnel" is evidence of CO2 absorption *and* emission, and water absorption and emission if you look more carefully at the plots. The earth's surface emits a spectrum which is probably a reasonably uniform black body shape at say 285 Kelvin. That would be just above the blue curve in the barrattbellamy plot if the atmosphere were to disappear suddenly. The greenhouse gases absorb regions of this spectrum, but they also emit in the same regions of the spectrum. The emitted photons can also be reabsorbed. As you go higher in the atmosphere though the concentration of greenhouse gases decreases and many of the photons emitted by the greenhouse gas escape to space. The depth of the "funnel" reflects in some way the fact that the average temperature of the levels from which the emission comes is lower than the surface temperature. If you look at the plot again the CO2 "funnel" is obvious because it is in the middle of the peak of the curve. But to the left of the curve, the spectrum is well below the 280K blue line, so there is also a H20 "funnel". And to the right another "funnel" for H20, CH4 and N2O is identified where, again, the spectrum dips well below the 280 line.  |
|
|
|
Post by radiant on Oct 15, 2009 16:33:41 GMT
No! And this is a key point. Noting that "funnel" was not a term I used, the "funnel" is evidence of CO2 absorption *and* emission, and water absorption and emission if you look more carefully at the plots. The earth's surface emits a spectrum which is probably a reasonably uniform black body shape at say 285 Kelvin. That would be just above the blue curve in the barrattbellamy plot if the atmosphere were to disappear suddenly. The greenhouse gases absorb regions of this spectrum, but they also emit in the same regions of the spectrum. The emitted photons can also be reabsorbed. As you go higher in the atmosphere though the concentration of greenhouse gases decreases and many of the photons emitted by the greenhouse gas escape to space. The depth of the "funnel" reflects in some way the fact that the average temperature of the levels from which the emission comes is lower than the surface temperature. If you look at the plot again the CO2 "funnel" is obvious because it is in the middle of the peak of the curve. But to the left of the curve, the spectrum is well below the 280K blue line, so there is also a H20 "funnel". And to the right another "funnel" for H20, CH4 and N2O is identified where, again, the spectrum dips well below the 280 line.  Ok 1. You are saying Tyndal is wrong that cooling water only emitts waters signature 'calorific rays'? 2. You are showing me a graph that is said by the page you got that from to be constructed from modtran using the us standard atmosphere where the us standard atmosphere does not contain water What actually is this pretty picture?? Do H20 emmissions end when C02's begin? no. Does the amospheric windows emmissions begin when C02 ends if C02 is absent. no. C02 only absorbs in a few lines. There is surely an overlap. What is the c02 contribution therefore? well surely less than the graph implies. What was the water concentration used to model this graph? Maybe there was so much 'noise' they had to reduce the water concentration to show us what they mean? Etc. Why on earth do you use this graph to discuss science when the page is so hopelessly inadequately documented? Tyndall used a simple cube filled with hot water with sides of gold silver copper and varnish to show that surface properties influence how a body radiates. Gold silver and copper being poor emitters compared to varnish. He said the metals generally were poor emitters. Are you agreeing with Kiwi that water is a black body emitter? A black body emitter does not have spectral peaks. it just has an infinite number of vibrations and wavelengths I know i need education in this whole area but it seems in major conflict with tyndall and i would have to dig up the references to what he did say and correct myself or present them here if you are saying that water does not emitt the spectra of water but is instead a black body emitter. |
|
|
|
Post by icefisher on Oct 15, 2009 16:57:02 GMT
Whether the amount is significant is a subjective matter. In my view, an apparently small increase of 3.7W/m^2 for a doubling of CO2 (which you'd hardly notice on the plot unless you measure it carefully) which may lead to 1.5-4.5C of surface warming *is* significant because in my subjective opinion I'd rather we didn't take such risks with our grandchildren's planet. Too bad you weren't around to regulate amoebas about a billion years or so ago. Something your grandchildren could not thank you for. Quite honestly Steve its pretty easy to fall into a religious rut. . . .and you certainly have here with that argument. There is no realistic alternative to progress. Al Gore is wrong with his sandpile theory. We need to conserve stuff but for the right reasons. Taking risks is what a successful life is all about. Testing the waters is necessary for learning. If you regulate regulate and regulate against learning you learn nothing. I recommend you pick up a copy of Hard Green by Peter Huber and give it a fair read. We are not far apart as you imagine but there is a deep chasm there. The premise is simple. . . .you react and change what is ugly, not what you think might be ugly. Risk taking is necessary for learning. Without learning we are not the master of our own world. That said. . . .it seems to me the 3.7watt number, and certainly the 1.5C to 4.5 C, only survives if both Kiwi and Hammer are wrong so perhaps we should try to stay on topic and discuss that. |
|
|
|
Post by steve on Oct 15, 2009 17:30:34 GMT
Where did I say that?
I've also pointed you to a link with an observed spectrum. Also, I note the barrattbellamy page15 shows an observed spectrum which is very similar. The plot is shown for illustrative purposes mainly.
The pretty picture is a spectrum. I suggest you plot it using MODTRAN without the CO2 to see what the overlaps are.
Because I am illustrating a broad point where minor inaccuracies in a graph don't matter. The broad point is that dips in the spectrum represent absorption and emission of greenhouse gases, they don't represent an *absence* of emission.
The emissivity of water and water vapour are very different surely? Water emissivity is high in the IR region - didn't you link to a plot of that. I don't quite see what the conflict with Tyndall is, but I've not looked in detail at your discussion with Kiwi.
|
|
|
|
Post by steve on Oct 15, 2009 17:48:43 GMT
Whether the amount is significant is a subjective matter. In my view, an apparently small increase of 3.7W/m^2 for a doubling of CO2 (which you'd hardly notice on the plot unless you measure it carefully) which may lead to 1.5-4.5C of surface warming *is* significant because in my subjective opinion I'd rather we didn't take such risks with our grandchildren's planet. Too bad you weren't around to regulate amoebas about a billion years or so ago. Something your grandchildren could not thank you for. Quite honestly Steve its pretty easy to fall into a religious rut. . . .and you certainly have here with that argument. No I'm just pointing out in my overly dramatic way your unscientific way of stating that something is "not significant". |
|
|
|
Post by radiant on Oct 15, 2009 19:57:09 GMT
my comments in yellowWhere did I say that? It was implied because Tyndall showed that ice water and water vapour all emit the same calorific rays. So that if you pass the rays of a hot electric lamp thru water so the lamps rays are so strong it almost boils the water (by the end of the experiment) the rays of the lamp leaving the water will pass thru ice without melting it while being able to warm a thermocouple on the other side of the ice
So the oceans absorb the suns rays to great depth but only emitt water vapour emission from the very top layer since water absorbs waters emissions in only 0.27inches of water.
Further tyndall knew that water finds it hard to create water vapour unless near water and unless warm which means that the vapour pressure of water is very very low down in the atmosphere unless forced upwards by convection or mountain currents. The warm oceans are therefore capped by a vapour that absorbs totally its own emissions and above a certain height these emissions have little ability to be absorbed by water and so can radiate to space at a relatively low altitude.
So i have to ask you again, was Tyndall wrong?
Tyndall also collected samples of the atmosphere from around the world and knew that central asia areas were cold not because of altitude but because of absense of water vapour. The pressure of water vapour being low where there is no water. Hence those dry deserts.
Tyndall seems to have some good insights into basic science few of us know about today. He makes a good read! For example freezing cold thermometer temperatures in the morning where there is no water in the interior of africa and yet the air being warm followed by exceptionally hot directly heated thermometer temperatures in the day.
Today science is reinventing and discovering what Tyndall already knew via the Nightcool studies to get cooler buildings by design using natural radiation. Also these kinds of studies show that even in antartica where it is very dry the skys temperature has more to do with water than any other substance
My theory that hose pipes and irrigation might play a greater role in global warming than c02 seems to be gaining credibility!
I've also pointed you to a link with an observed spectrum. Also, I note the barrattbellamy page15 shows an observed spectrum which is very similar. The plot is shown for illustrative purposes mainly.
Yes. Simulations with no clear explanation what is being simulated and for some reason a 1974 satellite view.
Why is it so impossibly difficult to find credible data?
Your nature reference may have been done over oceans to avoid 'noise'. 'Noise' which is just real data
The pretty picture is a spectrum. I suggest you plot it using MODTRAN without the CO2 to see what the overlaps are.
the pretty picture is a simulation. I recall now i did play with a tool provided by a well known faithist and when i looked at what i got i realise how the bellamy graph was made and i thought bullshit!! something to do with the extraordinary low level of water vapour in the air for the 'pretty picture' you want me to look at.
Then you accuse icefisher of being unscientific
Because I am illustrating a broad point where minor inaccuracies in a graph don't matter. The broad point is that dips in the spectrum represent absorption and emission of greenhouse gases, they don't represent an *absence* of emission.
What I think your graph shows is a simulation of terrestial black body emission with very low water vapour content being absorbed by c02. I suggest you play around with the controls until you produce that pretty picture and let me know the water concentration that produces it.
The emissivity of water and water vapour are very different surely? Water emissivity is high in the IR region - didn't you link to a plot of that. I don't quite see what the conflict with Tyndall is, but I've not looked in detail at your discussion with Kiwi. All i know is that tyndall says that ice water and water vapour all emitt the same calorific rays that are different to the rays emitted by other gases What actually do you mean by the emissivity of water and water vapour being different? They cant have the same absorbance because their concentration is different so you have to measure water vapour as an amount of precipital water
|
|
|
|
Post by steve on Oct 16, 2009 15:20:01 GMT
Icefisher started by saying there was no energy emitted, then energy *was* emitted at different frequencies, *then* energy was emitted but it was "not significant" which is like saying "I'm a bit pregnant". If you have a theory that the spectrum is completely unrepresentative then by all means present it. I admit I don't know how much water vapour is in this clear sky image, or whether and how the water vapour content would modify the plot. I haven't said Tyndall was wrong, I just haven't yet though enough about the different circumstance of looking at emissivity of water and water vapour. But I have some initial thoughts below based on a bit of digging around in a short amount of time. Feel free to dig more and help out. I went away and found a plot of "precipitable water". A measure of water vapour in the atmosphere. weather.unisys.com/upper_air/ua_con_prec.htmlThe answer seems to be up to a couple of inches (50mm) or so. So possibly the difference between the results for water and water vapour are related to the fact that there isn't that much water in the atmosphere  I also found a study of water vapour transmission from 1970 that indicated that there was a "water vapour window" which, if I've interpreted their graphs right, was relatively unsaturated for the sorts of amount of water vapour above (emissivity was 30% for 100mm) Journal of Applied Meteorology Flux Emissivity Tables for Water Vapor, Carbon Dioxide and Ozone D.O. Staley and G.M. Jurica 1970 As we do get infrared pictures of the earth from space, that doesn't sound unreasonable to me. What do you think? As a comparison, the same paper shows values of 20% for 1 metre of CO2 (385ppm is approx equivalent to 3.85 metres IIRC) mostly related to the 15 micron band (which is the position of the CO2 funnel in the barrattbellamy plot). If you accept that CO2 is evenly spread in the atmosphere (in proportion to atmospheric pressure) whereas much of the water vapour is below a few km of height, then that result could help explain the above plot. For example, half of the atmosphere is above 5500m or so (I worked that out when I was at 5500m, so my brain might have been a bit fuddled), so half of the CO2 is above this height. But a much smaller proportion of the water vapour is above such a height (I don't know how much). I've agreed it is hard to find web-accessible plots. I've pointed you to a much more recent paper with a more recent satellite image. I've said that it also contains a qualitatively similar CO2 feature. I guess you could look yourself and decide for yourself whether my interpretation is fair or wrong. |
|
|
|
Post by radiant on Oct 16, 2009 16:07:09 GMT
Icefisher started by saying there was no energy emitted, then energy *was* emitted at different frequencies, *then* energy was emitted but it was "not significant" which is like saying "I'm a bit pregnant". If you have a theory that the spectrum is completely unrepresentative then by all means present it. I admit I don't know how much water vapour is in this clear sky image, or whether and how the water vapour content would modify the plot. I haven't said Tyndall was wrong, I just haven't yet though enough about the different circumstance of looking at emissivity of water and water vapour. But I have some initial thoughts below based on a bit of digging around in a short amount of time. Feel free to dig more and help out. I went away and found a plot of "precipitable water". A measure of water vapour in the atmosphere. weather.unisys.com/upper_air/ua_con_prec.htmlThe answer seems to be up to a couple of inches (50mm) or so. So possibly the difference between the results for water and water vapour are related to the fact that there isn't that much water in the atmosphere  I also found a study of water vapour transmission from 1970 that indicated that there was a "water vapour window" which, if I've interpreted their graphs right, was relatively unsaturated for the sorts of amount of water vapour above (emissivity was 30% for 100mm) Journal of Applied Meteorology Flux Emissivity Tables for Water Vapor, Carbon Dioxide and Ozone D.O. Staley and G.M. Jurica 1970 As we do get infrared pictures of the earth from space, that doesn't sound unreasonable to me. What do you think? As a comparison, the same paper shows values of 20% for 1 metre of CO2 (385ppm is approx equivalent to 3.85 metres IIRC) mostly related to the 15 micron band (which is the position of the CO2 funnel in the barrattbellamy plot). If you accept that CO2 is evenly spread in the atmosphere (in proportion to atmospheric pressure) whereas much of the water vapour is below a few km of height, then that result could help explain the above plot. For example, half of the atmosphere is above 5500m or so (I worked that out when I was at 5500m, so my brain might have been a bit fuddled), so half of the CO2 is above this height. But a much smaller proportion of the water vapour is above such a height (I don't know how much). I've agreed it is hard to find web-accessible plots. I've pointed you to a much more recent paper with a more recent satellite image. I've said that it also contains a qualitatively similar CO2 feature. I guess you could look yourself and decide for yourself whether my interpretation is fair or wrong. The pretty picture is not a clear sky image. it is a simulation. It was you who suggested i could play with modtran to learn more. So you go learn about what created that graph since you dont know and learn more. Tyndall showed that water absorbs all the emissions of water from a oxyhydrogen flame producing only water vapour gas and unburnt fuel at 5898 F in a length of 0.27 inches of water and absorbs 97% in 0.04inch of water 'Only 2 inches of water' to you means a huge amount to Tyndall Even with very low amounts of precipital water in the entire atmosphere above antartica of the order of 1mm, water is the strongest source of all sky IR emissions. One metre of pure C02 is equivalent to 2597M of C02 in air. But you must know that from the 385ppm so i am not sure what you are saying with the 3.85m which is so similar numerically to the 385ppm I think you need to go learn about these things yourself rather than telling me i am wrong and me attempting to explain to you what has taken me hours of hard work to learn which you can find out for yourself from your own point of view so that you can counter what i am saying based on knowledge. I already pointed out that your recent study appears to be based on observing ocean based emissions without clouds because the other data was too 'noisy'. You began your reply with 'no' It seems you are unable to comprehend what i am talking about so far. Your models are based on a world that does not exist. The oceans are kept warm because of the nature of water but you seem clueless as to that nature and yet want to lecture other people how certain you are of the influence of C02 from a point of ignorance Everybody is learning. I think you need to catch up a bit If you measure the emission of water and water has a window then no radiation is emitted in the region of c02 that exists in the window. The only emission from C02 will be created by warming the trace amount of c02 in the warm atmosphere that can be absorbed in the colder atmosphere above. Icefisher said the C02 well was a result of no emission in that area. You said he was unscientific and began your reply to me with 'no' Your attitude does not create a discussion. You begin with your desired result and then work backwards to say what is necessary |
|
|
|
Post by steve on Oct 16, 2009 16:51:05 GMT
For the third time, I have seen a plot in a Nature paper from within the last 10 years that proves to me that the "CO2 funnel" exists. Forget about barrattbellamy and MODTRAN.
He also showed that enough heat passed through water to be measurable by a thermocouple. So there is a "window" in the spectrum where water and water vapour may not be so effective. That suggests to me that the assumption you've drawn from the above experiment is wrong. As does the fact that infrared telescopes can see the surface of the earth.
I'm talking about 1 metre of CO2 gas at standard temperature and pressure. If the whole atmosphere were at standard temperature and pressure it would be about 10km deep. Since CO2 is 385ppm by volume, that equates to 385/1000000*10000metres which is 3.85 metres at standard temperature and pressure. Might be a bit more. Might be a bit less. But 1ppm to 1cm is a nice easy number for my tiny brain to remember.
Answer the points. Obviously I think I know more about this than you do. And I've done my own "hours" of study. So lets just discuss questions, clarify things and understand that some of the issues we have are communication issues rather than straightforward disagreements.
I didn't say "No" to that bit, I said "No" to this bit: "so you would expect a funnel where water does not emit if by funnel we mean absense of emission or low emission?" (my bold). Do you stand by this statement? Because on the face of it it implies a fundamental misunderstanding of the nature of spectral lines.
|
|
|
|
Post by radiant on Oct 16, 2009 18:06:58 GMT
my reply in yellow For the third time, I have seen a plot in a Nature paper from within the last 10 years that proves to me that the "CO2 funnel" exists. Forget about barrattbellamy and MODTRAN. As i said many times the one you quoted may have been over water because of 'noise' when over ground because i found a similar study by one of the same authorsHe also showed that enough heat passed through water to be measurable by a thermocouple. So there is a "window" in the spectrum where water and water vapour may not be so effective. That suggests to me that the assumption you've drawn from the above experiment is wrong. As does the fact that infrared telescopes can see the surface of the earth. I am not sure where this water window is if water has high emissivity of .96, but if water does not emitt there, then C02 cannot absorb there if you measure over water.
And unless the sun is at the bottom of the ocean emitting radiation, that water does not absorb, then water cannot emitt other radiation. If you place a hot electric lamp one side of water, rays pass thru water and thru ice to a thermocouple. which rays pass thru? high energy rays or low energy rays
Water must be less transparant to the suns high energy infrared which is the principal of the greenhouse effect. Tyndall showed that Iodine dissolved in some subtance was totally opaque to sunlight so that a darkened room could have the heat rays of sunshine focused in total darkness to burn holes in metal foil and burn paper. The sun must emit IR which passes thru water to some depth? or water would steam at the surface in sunlight and be very warm at the very surface level - it isnt but it does get colder quite quickly with depth. But the radiation water emits most strongly is absorbed in almost no depth of water
Many ir cameras use high energy Ir at night to view 'invisible' objects. Visible light is high energy EMF.
So if water has a window and the studies measure over the oceans no upwards pointing energy of significance can be produced in those areas. Therefore Co2 will be warmed and emits via conduction and transmits radiation upwards so it cools the lower air slightly and sends some warmth back down but it cannot absorb emissions from the water at the c02 funnel because there are none or few.
Also even if water and Co2 areas appear to overlap it does not mean they overlap line by line.
I'm talking about 1 metre of CO2 gas at standard temperature and pressure. If the whole atmosphere were at standard temperature and pressure it would be about 10km deep. I think it will be lessSince CO2 is 385ppm by volume, that equates to 385/1000000*10000metres which is 3.85 metres at standard temperature and pressure. Might be a bit more. Might be a bit less. But 1ppm to 1cm is a nice easy number for my tiny brain to remember. Ok but I cant see where you get the 20% at one meter from though. Your paper seems theoretical rather than practical also. no actual measurements being made?Answer the points. Obviously I think I know more about this than you do. i seeAnd I've done my own "hours" of study. So lets just discuss questions, clarify things and understand that some of the issues we have are communication issues rather than straightforward disagreements. I think mostly you ignore what i say and assume you know better. and now you want to make out your are so reasonableI didn't say "No" to that bit, I said "No" to this bit: "so you would expect a funnel where water does not emit if by funnel we mean absense of emission or low emission?" (my bold). Do you stand by this statement? Because on the face of it it implies a fundamental misunderstanding of the nature of spectral lines. Perhaps you need to allow into your brain the fact you did not understand what i said before you dish out the arrogant clap trap |
|
|
|
Post by icefisher on Oct 16, 2009 20:43:51 GMT
Icefisher started by saying there was no energy emitted, then energy *was* emitted at different frequencies, *then* energy was emitted but it was "not significant" which is like saying "I'm a bit pregnant". There were no two "then"s. Its entirely what you imagined and you are jerk for not looking more carefully before impugning somebody with something they did not say. I replied directly: "Good catch! I should have said "no significant energy"." Hammer is the one making the case for the IR window and the thinning atmosphere in the regions where CO2 dominates may well turn what is significant when mixed with water vapor into insignificant when mixed with only CO2. Though I agree that for at least 2 seconds I went as stupid as you for assuming that N2 and O2 have no significant emission importance in the atmosphere. I agree that while sometimes its practical to assume absolutes, its pretty dangerous and stupid to jump to global systems and into quadrillions of tons of matter with experiments on pounds of matter. But that seems to be your stock in trade as you play top sycophant for the warmistas. However, the 2 seconds ended abruptly because ignorance is curable but stupidity is forever. |
|
|
|
Post by icefisher on Oct 16, 2009 21:19:17 GMT
He also showed that enough heat passed through water to be measurable by a thermocouple. Irrelevant! A 6,000 degree flame emits more than IR. So there is a "window" in the spectrum where water and water vapour may not be so effective. That suggests to me that the assumption you've drawn from the above experiment is wrong. As does the fact that infrared telescopes can see the surface of the earth. Ignorance at work! IR sensors do not have to "see" the surface. They register an IR signal that allows for computer programs to somewhat correctly render where the land is. Where it can't see the land the computer imposes a map. Land masses have bigger diurnal heat swings what the IR sensors are seeing is a heat signature and imprint if you will (like a tree ring sees temperature). When the water vapor gets thick enough to form clouds you can't even see the imprint. Where humidity gets low enough you might approach sensing the actual surface temperature. Like Duh!!!!!!!!!!! |
|
|
|
Post by radiant on Oct 17, 2009 4:56:17 GMT
He also showed that enough heat passed through water to be measurable by a thermocouple. Irrelevant! A 6,000 degree flame emits more than IR. So there is a "window" in the spectrum where water and water vapour may not be so effective. That suggests to me that the assumption you've drawn from the above experiment is wrong. As does the fact that infrared telescopes can see the surface of the earth. Ignorance at work! IR sensors do not have to "see" the surface. They register an IR signal that allows for computer programs to somewhat correctly render where the land is. Where it can't see the land the computer imposes a map. Land masses have bigger diurnal heat swings what the IR sensors are seeing is a heat signature and imprint if you will (like a tree ring sees temperature). When the water vapor gets thick enough to form clouds you can't even see the imprint. Where humidity gets low enough you might approach sensing the actual surface temperature. Like Duh!!!!!!!!!!!  This near-infrared photograph of the Earth was taken by the Galileo spacecraft at 6:07 a.m. PST on Dec. 11, 1990, at a range of about 1.32 million miles. The camera used light with a wavelength of 1 micron, which easily penetrates atmospheric hazes and enhances the brightness of land surfaces This near-infrared photograph of the Earth was taken by the Galileo spacecraft at 6:07 a.m. PST on Dec. 11, 1990, at a range of about 1.32 million miles. The camera used light with a wavelength of 1 micron, which easily penetrates atmospheric hazes and enhances the brightness of land surfacesFrom the following you can see I think that water is 10 times more opaque at 1 micron than for red light and then absorbs much more effectively in the mid and far infra red which ends at 100 micron. There being no absense of absorption in the infra red. Interestingly the sky is a deeper blue on humid days and water and snow appear blue because more red is absorbed as shown. omlc.ogi.edu/spectra/water/abs/index.html 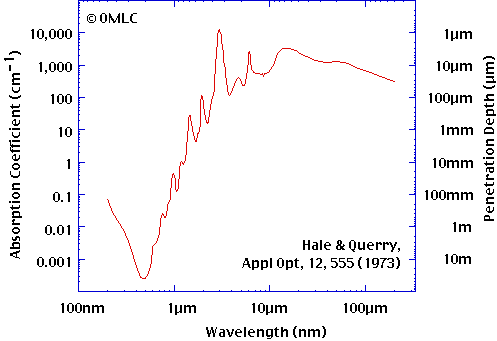 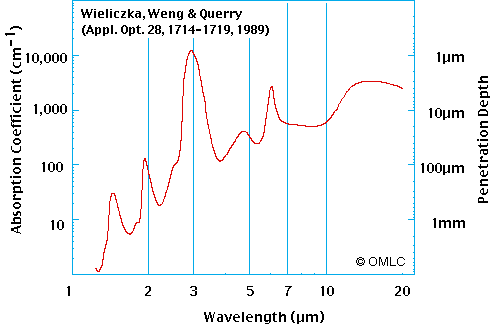 So at this point we need to step back and breathe deeply and look at that data i think The amazing quality of water is that it absorbs 100% all Ir even quite low in the low IR band from 1.8 micron and all of the mid and far band in only 1mm of water  Tyndall said that water absorbed 97% of the almost invisible Hydrogen flame emissions burning at 5898F in 0.04 of water or 1.016mm of water. An amazing tribute to the mans ability to measure the natural world  Nasas tribute to Tyndall: earthobservatory.nasa.gov/Features/Tyndall/he was able to express his thoughts in a manner none had seen or heard before. His ability to paint mental pictures for his audience enabled him to disseminate a popular knowledge of physical science that had not previously existed. Tyndall's original research on the radiative properties of gases as well as his work with other top scientists of his era opened up new fields of science and laid the groundwork for future scientific enterprises. |
|
|
|
Post by icefisher on Oct 17, 2009 6:40:56 GMT
From the following you can see I think that water is 10 times more opaque at 1 micron than for red light and then absorbs much more effectively in the mid and far infra red which ends at 100 micron. There being no absense of absorption in the infra red. Interestingly the sky is a deeper blue on humid days and water and snow appear blue because more red is absorbed as shown. 90% light blockage isn't much obstruction. A good pair of sunglasses can hit that level. Some snow goggles have only a 5% transmission rate and you wear them to see better in bright light conditions. |
|
|
|
Post by radiant on Oct 17, 2009 7:07:32 GMT
From the following you can see I think that water is 10 times more opaque at 1 micron than for red light and then absorbs much more effectively in the mid and far infra red which ends at 100 micron. There being no absense of absorption in the infra red. Interestingly the sky is a deeper blue on humid days and water and snow appear blue because more red is absorbed as shown. 90% light blockage isn't much obstruction. A good pair of sunglasses can hit that level. Some snow goggles have only a 5% transmission rate and you wear them to see better in bright light conditions. a 95% reduction in energy received is a huge difference The reason it seems like not much is because the human retina and brain has an amazing ability to compensate for different lighting levels even with no pupil size change so that humans have a very poor ability to gauge relative light levels. Human eye brain combination does not work like a video camera and computer monitor. The human eye has smarts in the retina to enhance what cannot be easily detected. There is also the more simple observation about snow goggles that they limit the light that we are least sensitive to and allow in an amount of the light we are most sensitive to which is yellow. The brain then probably compensates to make it seem less yellow based on clues from what other coloured light is detected Humans have a much poorer colour vision outside of the most central acute part of our vision. Eye movement and assumptions then fill in the parts we dont see properly to give us the illusion our eyes are able to see so fantasticly well but in reality this picture is constructed rather than simply received. |
|