|
|
Post by steve on Mar 27, 2009 7:54:30 GMT
I agree that the diagram is oversimplified because the enhanced greenhouse is a function of the profile of the atmosphere, and the diagram doesn't represent the profile of the atmosphere. That's why I put this question to Poitsplace: Essentially, what you are claiming is that the earth emits into space no radiation in the wavebands absorbed by CO2, because it has all been absorbed by the CO2. Do you believe that? No, I'm pointing out that because CO2 is stopping most of it's frequencies already...increasing it doesn't have the same affect. The system is already offloading MOST of the energy that CO2 absorbs. A great deal of the energy you seem to think is getting tied up in CO2...isn't. Poitspace. If even some radiation is being emitted by the earth in the wavebands that CO2 absorbs, then doubling the amount of CO2 will affect that radiation. You appear to agree with this, but you think the effect is small. I agree with you. The effect *is* small. By doubling CO2, the amount of radiation into space is predicted to reduce by only 2%. |
|
|
|
Post by steve on Mar 27, 2009 8:21:02 GMT
Warning: Cat amongst the pigeons: As I posted on the main global cooling thread, I want to point out (politely) that the entire atmosphere absorbs IR and radiates IR. Agreed, though most of the absorption/emission is actually done by the "greenhouse gases" within the atmosphere.Gases are not immune from the laws of thermodynamics and absorb & emit IR radiation. At the same temperature as the Earth surface, their emission signature is identical. The only thing different about "greenhouse gases" - a misnomer btw! - is that they can absorb and emit outside the blackbody curve. This effects the base blackbody signature, but doesn't upset the laws of thermodynamics. Not quite. Different materials have different emissivity. Nothing is a "black" body, but if you have two materials that are prone to emitting at different wavelengths (eg. greenhouse gases that emit/absorb IR and oxygen that emits/absorbs microwaves, then depending on the temperature of the atmosphere, the relative amounts of radiation from each will vary, and the spectrum will approximately fit under the black body curve for that temperature. I suppose if the atmosphere were very cold there would be much less radiation emitted overall, and a high proportion would be from the oxygen rather than from GHGs.Thus the WHOLE atmosphere participates in the greenhouse effect, allowing heat to move upwards in thermal equilibrium. ZERO heat can move back to the warmer earth surface (forbidden by the 2nd law of thermodynamics). The atmosphere is not in thermal equilibrium. In reality, a huge amount of latent heat is moved from the earth via water vapour to the atmosphere, where it condenses as heat, warming the atmosphere. Anytime the atmosphere is warmer than the surface, there will be a net thermal radiation back to the earth, being the difference between the two blackbody curves. I'm not saying that CO2 cannot have an effect. I'm saying that the classic greenhouse effect explanation is nonsense. Any blackbody radiation passing through molecules that absorb certain frequencies will absorb & re-emit. But any thermal, heat transfer, always, must, has to, obey the laws of thermodynamics. This is a jump too far. The temperature of the atmosphere is in part governed by the amount of radiation it absorbs. The radiation it absorbs is distributed to other molecules by collisions, and in turn CO2 molecules are sometimes excited by collisions following which they reemit. For a given amount of radiation, there may be net emission, or net absorption from the atmosphere depending on the temperature of the atmosphere. So you cannot say with such certainty that the CO2 has little effect |
|
|
|
Post by poitsplace on Mar 27, 2009 9:23:14 GMT
Warning: Cat amongst the pigeons: As I posted on the main global cooling thread, I want to point out (politely) that the entire atmosphere absorbs IR and radiates IR. Agreed, though most of the absorption/emission is actually done by the "greenhouse gases" within the atmosphere.Gases are not immune from the laws of thermodynamics and absorb & emit IR radiation. At the same temperature as the Earth surface, their emission signature is identical. The only thing different about "greenhouse gases" - a misnomer btw! - is that they can absorb and emit outside the blackbody curve. This effects the base blackbody signature, but doesn't upset the laws of thermodynamics. Not quite. Different materials have different emissivity. Nothing is a "black" body, but if you have two materials that are prone to emitting at different wavelengths (eg. greenhouse gases that emit/absorb IR and oxygen that emits/absorbs microwaves, then depending on the temperature of the atmosphere, the relative amounts of radiation from each will vary, and the spectrum will approximately fit under the black body curve for that temperature. I suppose if the atmosphere were very cold there would be much less radiation emitted overall, and a high proportion would be from the oxygen rather than from GHGs.Thus the WHOLE atmosphere participates in the greenhouse effect, allowing heat to move upwards in thermal equilibrium. ZERO heat can move back to the warmer earth surface (forbidden by the 2nd law of thermodynamics). The atmosphere is not in thermal equilibrium. In reality, a huge amount of latent heat is moved from the earth via water vapour to the atmosphere, where it condenses as heat, warming the atmosphere. Anytime the atmosphere is warmer than the surface, there will be a net thermal radiation back to the earth, being the difference between the two blackbody curves. I'm not saying that CO2 cannot have an effect. I'm saying that the classic greenhouse effect explanation is nonsense. Any blackbody radiation passing through molecules that absorb certain frequencies will absorb & re-emit. But any thermal, heat transfer, always, must, has to, obey the laws of thermodynamics. This is a jump too far. The temperature of the atmosphere is in part governed by the amount of radiation it absorbs. The radiation it absorbs is distributed to other molecules by collisions, and in turn CO2 molecules are sometimes excited by collisions following which they reemit. For a given amount of radiation, there may be net emission, or net absorption from the atmosphere depending on the temperature of the atmosphere. So you cannot say with such certainty that the CO2 has little effect The problem is that the relationship between the final numbers on CO2 absorption and the actual temperature increase is not its self linear either. It too...is logarithmic. The photons are captured and emitted again...and neither of those interactions actually increases temperature. As it repeatedly goes back and forth there's a HUGE chance that it will eventually (having never been heat) get shifted to water. Every time it hits water...odds are it's leaving in the wrong frequency. If it "thermalizes" odds are the black body radiation will emit in other frequencies as well. This too is a bit oversimplified, but... CO2 absorption is logarithmic...not good for capturing significant amounts more energy CO2 must compete with water vapor. The moment it loses, about 75% of the energy is pretty much gone. Most of the energy thought to be tied up bouncing back and forth between CO2 molecules actually ends up hitting water vapor eventually...and then it just leaves. The conversion of the additional, captured energy isn't linear...not entirely sure but it too is probably logarithmic. (granted in a better place along the curve than CO2 but still well into the point of diminishing returns)...not good for capturing more energy The rate at which any imbalance is radiated away is exponential...not good for capturing more energy when the very act of capturing makes radiating it away far more likely. LOL, Gore's "settled science" is writing checks that CO2 just can't cash. Since the concept is fundamentally flawed, the models are so far off they're considered falsified, the tell-tale heat signature is missing and the temperature hasn't gone up any (has actually gone down)...it's obvious that the environment is quite simply too insensitive to increases in CO2 for alarmist predictions to come true. If you'd like to go for a grand total of perhaps 1.5C for all forcing/feedbacks I'll let you have it just for the sake of being fair (and making it go qicker) but then you have to deal with my apathy on the whole issue. I really don't care much about an additional .5C or so by 2100 when I know the earth's been warmer during the holocene anyway? |
|
|
|
Post by glc on Mar 27, 2009 9:44:03 GMT
So, if the upper atmosphere is getting warmer, why has it shrunk by 100+ km? When I talk about the "top of the atmosphere" I'm referring, broadly speaking, to the tropopause, i.e. the region which separates the troposphere and stratosphere. The height of the troposphere is between ~7Km (at the poles) to ~20Km (in the tropics). The 'shrinkage' you mention refers to the ionosphere which extends more than 1000 Km into space. |
|
|
|
Post by glc on Mar 27, 2009 11:27:32 GMT
Why increasing CO2 might (and probably will) increase global temperatures.
Forget, for the moment, about saturation. Forget also about convection, conduction, evaporation etc. These are processes by which heat energy is transferred to the “Top of the Atmosphere” (TOA). At the TOA - radiation rules. It is by radiation from greenhouse gases that most of the ~235 w/m2 is emitted. Around 40 w/m2 is emitted from the earth’s surface and passes directly through the “IR window “. Right…..
Imagine now there are 300 ppm CO2, well-mixed throughout the atmosphere. Some of that CO2 will be near the surface and some will be much higher in the atmosphere (troposphere). At these higher levels the concentration of water vapour is very weak (see earlier discussion) so radiation emission will be dominated by CO2 (see emission spectra graphs). Note from the emission graphs that emission form the CO2 spectrum is at much colder temperatures than from water vapour. This reinforces an important point, i.e. temperature and altitude are linked. In the troposphere: Higher means Colder.
Ok - so we have 300 ppm CO2 absorbing and emitting at various levels of the troposphere. Now we have a second important point, i.e. energy (E) emitted is proportional to temperature raised to the 4th power (T4). This is a consequence of the S-B formula. But what happens if CO2 concentrations are raised from 300ppm to 600ppm?
Answer: More CO2 accumulates higher in the troposphere which means that more energy is emitted from higher levels.
So what?
Answer: Well we know it is colder at higher levels so energy will be emitted from a colder region. Also, from S-B, we know that temperature and energy are related, so the energy emitted from the colder regions (at 600 ppm) will be lower than that emitted previously (at 300 ppm).
What does this mean?
Answer: It means that the upper ‘layer’ of the troposphere will need to warm up to maintain the in=out energy balance. This will then have a corresponding impact on the lower layers of the atmosphere (troposphere) in order to maintain the “lapse rate” which will eventually impact on the surface.
What will the impact be?
I don’t know. If, as predicted, there is warming in the upper troposphere then it is perfectly possible that water vapour concentrations will increase because warm air holds more moisture than cold air which may enhance the greenhouse effect still further. This is what is known as positive feedback. But, there are all sorts of arguments, both for and against, on this issue and I'm not going into them now.
|
|
|
|
Post by icefisher on Mar 27, 2009 12:39:41 GMT
So, if the upper atmosphere is getting warmer, why has it shrunk by 100+ km? When I talk about the "top of the atmosphere" I'm referring, broadly speaking, to the tropopause, i.e. the region which separates the troposphere and stratosphere. The height of the troposphere is between ~7Km (at the poles) to ~20Km (in the tropics). The 'shrinkage' you mention refers to the ionosphere which extends more than 1000 Km into space. I get the "delay" idea in cooling for greenhouse gases. . . .but doesn't this cancel out from absorbing incoming blackbody radiation from the sun? With lots of humidity and clouds you get both less heating and less cooling. So why not with CO2 also? Why are CO2 models exclusively one-way streets? |
|
|
|
Post by steve on Mar 27, 2009 13:18:40 GMT
The problem is that the relationship between the final numbers on CO2 absorption and the actual temperature increase is not its self linear either. It too...is logarithmic. That's not a problem. Calculate the "forcing", which is essentially the outgoing longwave radiation calculated using an average atmosphere profile with 280 ppm CO2 compared with the same profile and 560ppm. Empirically, this is approx: 5.35 ln(Cnew/Coriginal) = 3.7 Watts/metre^2 for a doubling of CO2. Calculate the impact of this forcing (before feedbacks) assuming current outgoing longwave is about 235 W/m^2 (Tnew/Torig)^4 = 235/(235-3.7) Tnew = 287 Kelvin * fourth root of (235/231.3) = 288 Kelvin A 1C rise. No. More than 99.999% of the time a CO2 molecule which is excited loses its energy to a collision with another molecule. The GHG molecules each emit and absorb in the same frequency range, and also there is a lot of overlap between CO2 and H20. The idea of photons bouncing between CO2 molecules is wrong. Air is warm enough such that CO2 molecules are mostly excited and deexcited by collision. Where radiation is absorbed, it is quickly redistributed to other molecules to warm the atmosphere. But an excited molecule has a chance of emitting radiation which will cool the atmosphere if the radiation escapes. Note the above equation included the "exponential" bit. You keep suggesting the effect is small/logarithmic etc. and I'm in full agreement with that. But small is relative. 1C sounds small but may be significant. You're now shifting the goalposts a bit from your original post by adding the claim that it is "obvious" that the environment is insensitive. What is your evidence for this? It's obvious to *me* that the climate has swung through vast extremes in the past due to initially small kicks such as caused by slight variations in the earth's orbit and axis. Why would it be less sensitive now? |
|
|
|
Post by steve on Mar 27, 2009 13:31:05 GMT
So, if the upper atmosphere is getting warmer, why has it shrunk by 100+ km? When I talk about the "top of the atmosphere" I'm referring, broadly speaking, to the tropopause, i.e. the region which separates the troposphere and stratosphere. The height of the troposphere is between ~7Km (at the poles) to ~20Km (in the tropics). The 'shrinkage' you mention refers to the ionosphere which extends more than 1000 Km into space. I get the "delay" idea in cooling for greenhouse gases. . . .but doesn't this cancel out from absorbing incoming blackbody radiation from the sun? With lots of humidity and clouds you get both less heating and less cooling. So why not with CO2 also? Why are CO2 models exclusively one-way streets? Most of the radiation from the Sun is in the visible region. Clouds can reflect this radiation away, but CO2 and water vapour is transparent to it, so if the sky is clear, much of it gets through to be absorbed by the surface. Most of the radiation from the earth's surface or atmosphere is in the infrared region much of which is absorbed by greenhouse gases such as water vapour and CO2. That's why it's a "1 way street". For parts of the spectrum it isn't a one-way street. Eg. UV is absorbed by ozone in the upper atmosphere resulting in warming of the stratosphere. Extra greenhouse gases here actually help to cool the stratosphere because the extra molecules mean more radiation is emitted, but the thin atmosphere means the radiation can escape to space. |
|
|
|
Post by icefisher on Mar 27, 2009 17:53:42 GMT
I get the "delay" idea in cooling for greenhouse gases. . . .but doesn't this cancel out from absorbing incoming blackbody radiation from the sun? With lots of humidity and clouds you get both less heating and less cooling. So why not with CO2 also? Why are CO2 models exclusively one-way streets? Most of the radiation from the Sun is in the visible region. Clouds can reflect this radiation away, but CO2 and water vapour is transparent to it, so if the sky is clear, much of it gets through to be absorbed by the surface. Most of the radiation from the earth's surface or atmosphere is in the infrared region much of which is absorbed by greenhouse gases such as water vapour and CO2. That's why it's a "1 way street". That doesn't seem to be an explanation. I understand that the sun's blackbody radiation includes light that is visible, thats because its a stronger radiation source. If the earth were the temperature of the sun it would radiate at the same frequencies as the sun. I have heard arguments as to how it delays the reemission of the IR providing a period of time where the planet warms; but would not such delays also operate on incoming radiation blocking it, absorbing it, accumulating it, holding it and reradiating to non-sun occupied areas of space during the daytime and during the evening time work fulltime to do the same from surface IR? A physical process that delays the cooling would also serve to delay the heating from sun IR in the same spectrum as well. |
|
|
|
Post by tacoman25 on Mar 27, 2009 18:42:00 GMT
No, no, a thousand times no! CO2 and other greenhouse gases only stop a portion of the energy going into space and re-radiate a fraction of that portion back down toward the surface. However, it's enough to warm the planet by 33 degrees. Here's a scematic diagram of the energy flows in the climate: 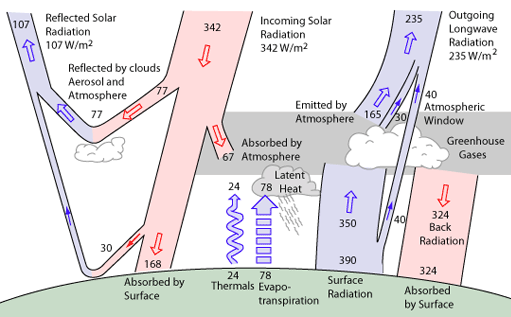 And here's a paper to read on energy flows in the climate system: ams.allenpress.com/archive/1520-0477/preprint/2008/pdf/10.1175_2008BAMS2634.1.pdfThe problem that some people on here seem to have is that they don't realize some, like myself, CAN BELIEVE IN THE GREENHOUSE EFFECT IN GENERAL, but not be entirely convinced that increases in CO2 alone are causing such an enhanced greenhouse effect as the IPCC and others believe. It is indeed possible to be doubtful that CO2 feedbacks are enough to cause such temperature rises, without doubting the fact that greenhouse gases keep the earth much warmer than it would be otherwise. Believing in the greenhouse effect, does not mean one has to buy into AGW theory. |
|
|
|
Post by steve on Mar 27, 2009 18:42:38 GMT
Icefisher,
Maybe I'm not understanding your question.
You seemed to be saying that increasing CO2 would reduce the amount of energy getting to the surface (like clouds do).
|
|
|
|
Post by socold on Mar 27, 2009 19:37:04 GMT
Believing in the greenhouse effect, does not mean one has to buy into AGW theory. Yes exactly! I should have put it like this days ago |
|
|
|
Post by icefisher on Mar 27, 2009 20:16:27 GMT
Icefisher, Maybe I'm not understanding your question. You seemed to be saying that increasing CO2 would reduce the amount of energy getting to the surface (like clouds do). Sorry I was in a hurry to get to a meeting so I posted too soon. I reworded the question above so its clearer. Bottom line is I would like to understand the theory but am having trouble. The oceans operate in a similar manner diffusing energy throughout a variation of depths. Net result is a moderation of climate, not a net increase or decrease. The only thing different with the atmosphere would seem to be the gas laws of how temperature and pressure are interrelated providing a handy explanation for why the surface is warmer than the blackbody equilibrium temperature of the earth at the average terminal emission point up in the atmosphere. Is there any need for a greenhouse effect at all to explain the average temperature of the earth if we can correctly model all the albedo, convection, and radiation effects? Perhaps I am all wet on this. But real greenhouses diminish incoming radiation also to some degree. One has to wonder how you could calculate an inbalance, other than measure it some as it varies between incoming and outgoing. |
|
|
|
Post by bsattu on Mar 27, 2009 20:39:19 GMT
What about the tropopause? Inversion layer. Does the CO2 get stuck there?
|
|
|
|
Post by jimg on Mar 27, 2009 22:53:43 GMT
glc: dang units, I actually meant miles, my apologies: in metric units it's more like 200 km. which is not insignificant. (space boundary is 20% closer) Nevertheless, it's still a cooling issue. www.sciencedaily.com/releases/2008/12/081215121601.htmCINDI's first discovery was, however, that the ionosphere was not where it had been expected to be. During the first months of CINDI operations the transition between the ionosphere and space was found to be at about 260 miles (420 km) altitude during the nighttime, barely rising above 500 miles (800 km) during the day. These altitudes were extraordinarily low compared with the more typical values of 400 miles (640 km) during the nighttime and 600 miles (960 km) during the day. |
|