|
|
Post by kiwistonewall on Apr 1, 2009 11:42:05 GMT
Let's now look at the "greenhouse gases". The following diagram (upper) shows the claimed Earth's radiating temperature. (255K = -18C). In reality, there will be a number of such curves, with the peak displaced relative to the temperature of the radiating object. The lower diagram shows the absorption of a vertical beam of IR through the atmospheric gases. You can't relate the two directly, since in reality the Earth radiates in all directions upwards. Water & CO2 will also emit in all directions. All we can say is that IR will be absorbed and emitted many, many times. (BTW, this absorption is perfectly elastic, and the quanta of energy isn't changed by absorption & emission. Newton's Laws do NOT apply for the very small. We are at the level of quantum mechanics. There will be some thermalisation. There will also be thermal heat re-exciting molecules which will then re-emit. Since water is more abundant, it will 'capture' some of the energy absorbed by CO2, which is the cause of the CO2 bite in earth emission spectra. Much of the IR emitted from the Earth will pass directly to space. The atmosphere will absorb more effectively in the greenhouse gas regions, but these are a small percentage of the spectrum passing outward. In effect, the greenhouse gases are more efficient at BOTH radiating and emitting in their narrow spectral bands. But the overall result is the same - radiation passes up through the atmosphere being absorbed and emitted, but only temperature differences are important in determining the overall heat transfer. So both the theoretical thermodynamic calculations, and the experimentally observed spectrum of the Earth itself (still overall a black body curve) show us that normal thermodynamics is in play, and there is no thermodynamic distortion by "greenhouse gases"  Text with the above illustration: Figure 4. The radiation absorption characteristics of water vapour and carbon dioxide as a function of wavelength. The upper portion of the chart shows the wavelength distribution of radiation emitted from black bodies radiating at 6000K (approximately the solar photosphere) and 255K (approximately the earth’s planetary temperature), with the solar irradiance measured at the mean distance of the earth from the sun. The percentage absorption of a vertical beam by representative atmospheric concentrations of water vapour (H2O) and carbon dioxide (CO2) are shown in the lower panelsfrom: www.bom.gov.au/info/climate/change/gallery/4.shtml |
|
|
|
Post by steve on Apr 1, 2009 15:05:07 GMT
Kiwistonewall, You've misunderstood the plot. It says it is from a black body with the earth's temperature. It is not the earth's spectrum:  If you double CO2 you get more emission and more absorption throughout the atmosphere. At higher parts of the atmosphere though, some of the emission that used to get to space will now be absorbed, and the earth will need to depened on extra emission from slightly higher layers to re-achieve energy balance. But the troposphere is not in thermal equilibrium as it gets colder as you go up. Because the higher layers are colder, the extra emissivity caused by the extra CO2 doesn't make up for the reduced emission caused by the cooler temperature. If you do the calculations, you need these layers to warm by about 1C to re-achieve balance. As an aside, do you care about the fact that Lindzen and many other "sceptic" scientists disagree with you? |
|
|
|
Post by glc on Apr 1, 2009 15:21:09 GMT
Kiwistonewall,
You've misunderstood the plot. It says it is from a black body with the earth's temperature. It is not the earth's spectrum: Yes - I suspected he was misinterpreting it before, but gave the benefit of the doubt. I've tried to explain the plot and the higher cooler argument about half a dozen times but with little succes. Perhaps you'll have more luck. Kiwi: Though it hurts me to refer you to a 'warmer'  , read through Steve's post. He has it about right. |
|
|
|
Post by gridley on Apr 1, 2009 15:36:43 GMT
poitsplace, thank you for providing that image. Socold, I'm afraid that I don't consider a ~5% variation to be "well mixed"; especially since this is the variation in the troposphere, which would imply that the variation near the surface is measurably greater. Further, note where the concentrations are highest; aside from a spot in South America (looks to be around Buenos Aires?), the high concentrations are in the western US, the Atlantic off the US eastern seaboard, the Mediterranean, the middle east, and... it looks to me like Mongolia? This appears to me to correlate very poorly with GISS Temp data for a bounding period. For that matter, I see only a moderate correlation with human industrial zones (postulating that high CO2 concentrations should be 'downwind' of major industrial areas). Also, related to the above, if the ~104ppm anthropogenic increase in CO2 is so critical, how can a variation of 18ppm (roughly 17%) not be relevant? Finally, I noted in the text accompanying the image that "Climate modelers are currently using the AIRS data to understand the global distribution and transport of carbon dioxide and improve their models." Can anyone provide more data on the extent to which this uneven distribution is accounted for in GCMs? It appears that they are at least aware of it and trying to incorporate it as of 2003. Thanks all, Gridley Gridley Notice the date on the jpg that was supplied it is 2003. For some reason there is very little discussion of the magic gas CO2 other than it is always well mixed, which disagrees with the satellite image. No it doesn't, look at the scale. |
|
|
|
Post by ron on Apr 1, 2009 16:33:11 GMT
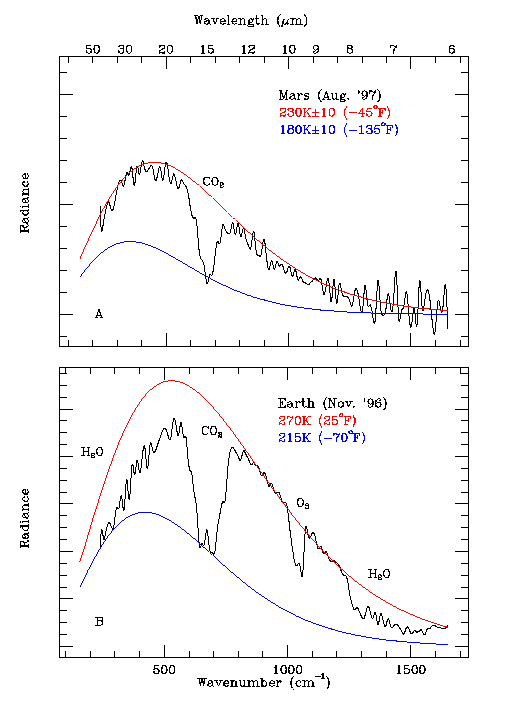 Odd that the shape of the "bite" differs for the same molecule. I wonder if there's an overlap going on? I wonder if one is much smaller, further away, has a less dense atomspher, etc, etc. No, we're talking about wavelength absorption. It should be the same shape every time for the same molecule. The intensity of the bite might change, but the SHAPE must remain the same. Otherwise things like spectroscopy wouldn't work. IMNSH,NSBO There is considerable overlap between water and CO2 absorption bands.
Exactly my thoughts. |
|
|
|
Post by socold on Apr 1, 2009 19:08:27 GMT
The reason convection carries away more heat is that backradiation is so strong. If there was no backradiation, radiation would transfer about 390wm-2, which easily overwhelms convection. So, why is the moon, during daylight, so much hotter than the surface of the earth? Anything in particular I said suggest that it shouldn't? |
|
|
|
Post by socold on Apr 1, 2009 19:29:03 GMT
As I sit here looking out over a swimming pool and a lake steaming in the pale dawn light ... I think I would refute your contention that only radiation from the surface can take heat from the surface. I said changing convection won't alter the rate of heat lost from the Earth (the planet), not the surface. Where have you looked? That's not an argument. Any numbers you come up with will be numbers accepted in the climate field right? In which case they are in the models. And again. If these numbers are accepted in the climate field then the models already encompass them. So you disagree with me that temperature will keep dropping without sunlight? Even with no feedbacks doubling co2 causes a very significant 1C global warming. I doubt it. Just as it's admitted the likely range of warming from doubling co2 is between 1.5C and 4.5C. That big range encompasses uncertainty with clouds. Even if we take the lowest value from that range that's still significant warming. |
|
|
|
Post by socold on Apr 1, 2009 19:31:24 GMT
The reason convection carries away more heat is that backradiation is so strong. If there was no backradiation, radiation would transfer about 390wm-2, which easily overwhelms convection. Have you got a cite for 'back radiation' from CO 2 its mechanism and strength? I am going off the energy budget figures |
|
|
|
Post by socold on Apr 1, 2009 19:46:35 GMT
Back scatter: No such thing from a thermodynamic standpoint.If the atmosphere is warmer than the ground, it will radiate downwards There's your back scatter. Radiation downwards from the atmosphere is a good description of "back scatter". The atmosphere won't radiate in a black body curve, because the atmosphere is not a backbody. Greenhouse gases emit IR (by definition), other gases do not because they don't vibrate to interfere with the electromagnetic field to produce IR light. The blackbody formulas assume a perfect blackbody. There is no mathematical formula for the atmosphere (which is not a blackbody) because the emission spectrum is very complex, although I think it can be derived from quantum physics nowadays. It isn't a simple curve. Moderately complex climate models contain "radiation code" in order to calculate the radiation propergation through the atmosphere for a reason. Why do radiative transfer codes ( en.wikipedia.org/wiki/List_of_atmospheric_radiative_transfer_codes) exist if IR emission can be simply calculated from temperature? There is a bite in the H2O spectrum, but due to multiple reasons it's not as obvious as the co2 one: - The H2O region is wide - The H2O IR emission is nearer the surface For these reasons it's less distinguishable from other regions where neither h2o or co2 absorb strongly (surface emission). The reason the co2 bite (and the ozone bite) is prominent is that: - They are narrow regions - Their emission comes from higher in the atmosphere (cooler) so the energy emitted into space is less (ie the bite). Aren't you at all bothered that all the experts haven't spotted your simple argument? They can't be that ignorant to miss somethign so obvious, so you must be proposing they are all liars. This reminds me a lot of Young Earth Creationists trying to explain to me why Geologists worldwide have missed their stunning argument against an earth older than a few thousand years old. |
|
|
|
Post by jimg on Apr 1, 2009 19:50:30 GMT
socold.
I'm still wondering if you are going to answer the question about water vapor?
You had previously implied that water vapor was at saturation.
Is this what you believe? Sources?
|
|
|
|
Post by socold on Apr 1, 2009 20:03:43 GMT
Socold, I'm afraid that I don't consider a ~5% variation to be "well mixed"; especially since this is the variation in the troposphere, which would imply that the variation near the surface is measurably greater. The variation near the surface is greater, but the well mixed label applies to the troposphere as a whole (of which the near surface is a minor part) Here is a gas that is not well mixed for comparison:  The co2 variations are too minor to expect any significant temperature variations. co2 is well mixed in the troposphere. It's spread out quite nicely overall so you can't easily see where it is coming from. The 17% variation is across a single month. I don't know if it would be possible (or correct) to compare that with temperature data for that month (itself containing a lot of variation) and discern the co2 signal. Perhaps over lots of months. Then again temperature and the co2 variations are always moving. The 104ppm rise (33%) is an increase in the annual mean. No idea, perhaps they want this to refine the carbon cycle models within the GCMs. |
|
|
|
Post by questioneverything on Apr 1, 2009 22:28:30 GMT
|
|
|
|
Post by kiwistonewall on Apr 2, 2009 3:10:21 GMT
I'm right & anyone who disagrees with me is wrong! (Proof by assertion & arrogant assumption!) Well it works for Hansen & Gore. Here is a tropical Earth emission: Note the absence of a H20 bite, but a peak instead. (H20 Window) The more abundant H20 scavenges energy absorbed by CO2 - this is a spectral anomaly and has no effect on the radiative outcome. For easy of viewing, cut & past image onto a white background!  1. Radiation is a property of ALL matter. So the greenhouse effect works without greenhouse gases!! (FACT) 2. Radiation transfer formula ONLY depend on Temperature & Temperature differences (FACT) 3. Heat cannot travel from Cold matter to warmer matter - so there is NO BACKSCATTER - that is imaginary - only thermodynamics applies. The warmer surface radiates to the cooler surface. This is a net effect & fully built into the thermodynamic equations. You can't stand in front of an electric bar heater and backscatter heat to the red hot bar! In reality there IS radiation from you toward the bar heater, but it can't be observed & it is entirely irrelevant. There is no way that your energy can mathematically effect the heater using any accepted thermodynamical equation. The so called greenhouse gases and the myth of greenhouse gas warming predates Thermodynamics (FACT) and isn't real. Fact- If the CO2 greenhouse theory was correct, then perpetual motion, self warming ovens and a whole lot of other nonsense follows! FACT. I know it is hard to get rid of a false paradigm. It takes years of patience from those who understand the truth. |
|
|
|
Post by kiwistonewall on Apr 2, 2009 3:14:56 GMT
Socold, you said:
"The atmosphere won't radiate in a black body curve, because the atmosphere is not a backbody. Greenhouse gases emit IR (by definition), other gases do not because they don't vibrate to interfere with the electromagnetic field to produce IR light."
That is wrong - Now you are saying that the Sun doesn't give off any heat!!!! Yes, that is what you are saying. The Sun is a giant ball of gas, giving off the blackbody radiation appropriate for its temperature. I guess all the warming comes from CO2 and the Sun is unnecessary? ;D
I've posted BB radiation curves from cold interstellar gases. BB radiation is a property of matter, and follows from the Boltzmann distribution - I've posted informative links to Physics sites in the past for those who want to see the truth.
|
|
|
|
Post by icefisher on Apr 2, 2009 5:57:27 GMT
But the troposphere is not in thermal equilibrium as it gets colder as you go up. Because the higher layers are colder, the extra emissivity caused by the extra CO2 doesn't make up for the reduced emission caused by the cooler temperature. If you do the calculations, you need these layers to warm by about 1C to re-achieve balance. Because it gets warmer as you go up? How much higher can you go? Just curious but it appears you have about an 80 degree drop between your water curve and your CO2 curve. Doesn't that already put CO2 emission at the top of the troposhere? I suppose if you add enough CO2 it might raise the stratoshere. |
|




 , read through Steve's post. He has it about right.
, read through Steve's post. He has it about right. 


