|
|
Post by icefisher on Aug 28, 2013 15:51:49 GMT
Could the heat of fusion cause a burst of 750watt/m2? If not why not? Please show your calculations! You need to learn to read! There is no such thing as heat of fusion of ice. Hmmmm, where did you read the word ice? You imagin'n sheet? |
|
|
|
Post by Andrew on Aug 28, 2013 16:00:13 GMT
You need to learn to read! There is no such thing as heat of fusion of ice. Hmmmm, where did you read the word ice? You imagin'n sheet? There is no such thing as the heat of fusion of water |
|
|
|
Post by magellan on Aug 28, 2013 16:10:05 GMT
Hmmmm, where did you read the word ice? You imagin'n sheet? There is no such thing as the heat of fusion of water |
|
|
|
Post by Andrew on Aug 28, 2013 21:08:44 GMT
After getting bored waiting for a response from NSIDC/NASA when it is fairly clear they do not understand the point that is being made, i decided to make it clearer what i was objecting to.
---------------------
I have been reviewing the responses I received from yourself and NSIDC.
It is fairly clear to me that you totally do not understand the point i am making. I am not making a point about one sentence in that article but rather the entire direction the article is moving in.
If you plot the temperature of cooling water and then icey water and then ice as a single graph as water cools you then get a measure of the heating ability of the different phases and mixtures of phases to heat the atmosphere. What you will see is there is a greater ability to heat the atmosphere prior to ice formation.
It is fairly clear to me that for some reason NSIDC believes that when ice forms that an additional source of heat is provided that has some ability to heat an atmosphere above it, more than water at 0.00000000000001C (for pure water) could do.
The fact is, there is no additional source of heat once ice formation begins and in any case as you pointed out, the ice formation reduces the ability of the water to release heat because the ice insulates the water from the cold atmosphere
Latent heat of fusion of water is a hidden heat. It has no ability to heat anything other than the heat that OC water has an ability to things.
What you find with latent heat of fusion, is that over a period of time there is more ability to heat the atmosphere that cools the water and water ice mixture because water has a latent heat of fusion, but in terms of heat per unit of time when ice forms there must be less heat to heat the atmosphere that cools the mixture, than the heat available per unit of time that was available prior to ice formation.
That article as it is currently written cannot be saved without a fairly major rethink of what was causing the unusual conditions at that time. I suspect for example that a mass of warm dry air moved in and the water radiated thru it to space and quickly cooled to enable the freezing, with the higher air temperatures not being directly related to the water freezing. Something like that.
Best Regards
Andrew
|
|
|
|
Post by Andrew on Aug 28, 2013 22:04:13 GMT
Reply from my man at NASA within the hour
------------------
Yes, I think I see your point. The wording implies that the rapid ice growth caused the warm temperatures, whereas in reality both the rapid ice growth and the warm temperatures were due to the large area of open water that needed to cool after absorbing a significant amount of heat. I passed along your comments to NSIDC with thoughts on how to better phrase the text to be clearer and technically correct
-----------------
|
|
|
|
Post by icefisher on Aug 28, 2013 23:48:12 GMT
good job andrew. you didn't try to sell numno's suggestion that zero heat rises.
i still suspect that the freezing process of saltwater involves some warming like we see with the supercooled water. Since the salt is squeezed out as the ice freezes and only gets trapped in pockets of brine in the ice the heat of fusion can warm the supercooled fresh water to 0C without stopping the freezing process.
You should have at least asked that question but I suspect they will probably keep it in as they have the reverse function clearly figured into why the summertime arctic is cooler. And of course no doubt some of the heat of fusion goes into sublimnation that cools the ice because the heat of sublimnation is several times that of freezing.
|
|
|
|
Post by Andrew on Aug 29, 2013 3:58:44 GMT
good job andrew. you didn't try to sell numno's suggestion that zero heat rises. i still suspect that the freezing process of saltwater involves some warming like we see with the supercooled water. Since the salt is squeezed out as the ice freezes and only gets trapped in pockets of brine in the ice the heat of fusion can warm the supercooled fresh water to 0C without stopping the freezing process. You should have at least asked that question but I suspect they will probably keep it in as they have the reverse function clearly figured into why the summertime arctic is cooler. And of course no doubt some of the heat of fusion goes into sublimnation that cools the ice because the heat of sublimnation is several times that of freezing. Numerouno was totally correct. There is no heat spike or "bursts of energy" coming from freezing water. Warming by supercooling provides no net heat because heat has to be extracted first to create the super cooling There is no such thing as the heat of fusion of water. (unless you are successfully able to supercool water without freezing it till it does eventually freeze, whereupon the mixture warms to be the temperature of whatever it would have been without the temporary delay in freezing for a totally zero sum ability to heat the mixture) |
|
|
|
Post by Andrew on Aug 29, 2013 6:34:07 GMT
Reply from my man at NASA within the hour ------------------ Yes, I think I see your point. The wording implies that the rapid ice growth caused the warm temperatures, whereas in reality both the rapid ice growth and the warm temperatures were due to the large area of open water that needed to cool after absorbing a significant amount of heat. I passed along your comments to NSIDC with thoughts on how to better phrase the text to be clearer and technically correct ----------------- Seems to me the writer is still trying to save that article! Why does he think he sees my point??? He says: "In reality the rapid ice growth and the warm temperatures were due to the large area of open water that needed to cool" This is horribly confused. If there was a rapid ice formation the open water had already cooled. Therefore when you correctly express part of what he has said you come up with: "In reality the rapid ice growth and the warm temperatures were due to the large area of open water that had already cooled" and obviously that makes no sense whatsoever unless you add in the observation that when ice freezes the atmosphere becomes colder because the warm water under the ice is no longer able to heat the atmosphere at the same rate, because the surface of the ice is far colder than the water.  |
|
|
|
Post by numerouno on Aug 29, 2013 9:48:17 GMT
good job andrew. you didn't try to sell numno's suggestion that zero heat rises. Zero spontaneous heat rises from the freezing. The heat that is moved elsewhere during the freezing is quite ordinary heat that we will be totally unable to tell apart from the other heat that is also being removed from the freezing body during the cooling. It's only that the cooling will take a bit longer to achieve than if that cooling was done with water only, owing to the latency involved with forming the ice. So, there will be no "spikes of energy"! |
|
|
|
Post by icefisher on Aug 30, 2013 5:18:13 GMT
good job andrew. you didn't try to sell numno's suggestion that zero heat rises. Zero spontaneous heat rises from the freezing. The heat that is moved elsewhere during the freezing is quite ordinary heat that we will be totally unable to tell apart from the other heat that is also being removed from the freezing body during the cooling. It's only that the cooling will take a bit longer to achieve than if that cooling was done with water only, owing to the latency involved with forming the ice. So, there will be no "spikes of energy"! Hmmmmmm, looks like Numno is easing over to the sensitive heat camp shuffling his feet sideways. Now the heat can rise to delay cooling. Indeed that is the process that keeps crops for getting too cold water at zeroC!!!!!!! Its getting cozy around here! LOL! This is a lot different than maintaining the heat remained hidden and was fully redeployed in the expansion of ice crystals. OK at least that is settled. We still have to deal with the warming of supercooled water. I would suggest that if seawater requires a temperature of -2C because of the salt content to freeze that the water is in fact supercooled. And if in the process of freezing the salt is squeezed out of the water, the water will have the ability to warm. Evidence? Pretty sketchy. But a strong indicator is that the arctic has been relatively cool all the time that ice has been melting. there needs to be an explanation for that. NSIDC says its from the absorption of sensitive heat into a latent form as the ice melts. Obviously if thats the case then there needs to be a reversal of that process as otherwise water would be a mysterious vessel into which energy poured and never was seen again, sort of a black hole or something like that. Of course its kind of hard to explain this year with the idea the cold summers were caused by melting ice. |
|
|
|
Post by Andrew on Aug 30, 2013 5:32:15 GMT
Zero spontaneous heat rises from the freezing. The heat that is moved elsewhere during the freezing is quite ordinary heat that we will be totally unable to tell apart from the other heat that is also being removed from the freezing body during the cooling. It's only that the cooling will take a bit longer to achieve than if that cooling was done with water only, owing to the latency involved with forming the ice. So, there will be no "spikes of energy"! Hmmmmmm, looks like Numno is easing over to the sensitive heat camp shuffling his feet sideways. Now the heat can rise to delay cooling. Indeed that is the process that keeps crops for getting too cold water at zeroC!!!!!!! Its getting cozy around here! LOL! This is a lot different than maintaining the heat remained hidden and was fully redeployed in the expansion of ice crystals. OK at least that is settled. We still have to deal with the warming of supercooled water. I would suggest that if seawater requires a temperature of -2C because of the salt content to freeze that the water is in fact supercooled. And if in the process of freezing the salt is squeezed out of the water, the water will have the ability to warm. Evidence? Pretty sketchy. But a strong indicator is that the arctic has been relatively cool all the time that ice has been melting. there needs to be an explanation for that. NSIDC says its from the absorption of sensitive heat into a latent form as the ice melts. Obviously if thats the case then there needs to be a reversal of that process as otherwise water would be a mysterious vessel into which energy poured and never was seen again, sort of a black hole or something like that. Of course its kind of hard to explain this year with the idea the cold summers were caused by melting ice. Icefisher you are totally unable to understand what is being explained to you. There is a limit to what another person can do to explain what you either cannot understand or you refuse to understand. Numerouno is totally correct. No heat rises to delay cooling. No heat per unit of time whatsoever is produced by freezing water that is not already fully available and produced by the heat of water per unit of time. No warming energy whatsoever per unit of time can be produced from freezing pure water at 0C, that is not already available in unfrozen water that is at 0.0000000000000000000001C Supercooling is a red herring. >>sensitive heat camp >>absorption of sensitive heat The word used in science is sensible. Meaning something that can be felt or experienced or sensed. |
|
|
|
Post by icefisher on Aug 30, 2013 6:29:58 GMT
Numerouno was totally correct. There is no heat spike or "bursts of energy" coming from freezing water. Warming by supercooling provides no net heat because heat has to be extracted first to create the super cooling There is no such thing as the heat of fusion of water. (unless you are successfully able to supercool water without freezing it till it does eventually freeze, whereupon the mixture warms to be the temperature of whatever it would have been without the temporary delay in freezing for a totally zero sum ability to heat the mixture)You mean I really have to take hot water and cool it before it will freeze and release energy? Imagine that!!! LOL!!!! the discussion was about this chart: (the blue line is 0C) As one can see, and it is seen every year the ocean water cools to about -2C.  So regarding the upward blip I said: this is the time of year when you start seeing heat spikes in the atmosphere from the release of water heat energy as the water freezes. (by time of year it meant when its below 0C)(by upward blip we are looking at slightly more than a 1 deg C burst of energy on the DMI data collectors) this is what Numno said was the fate of the energy: Of course no such thing will happen, the phase change energy will go into changing the water into the crystal structure of ice, which will also expand a bit. (And by which way nature chops down northern rocks and mountains chip by chip.)So Numno said the energy will be used to chop down mountains. Do you agree with that Iceskater? Seems Numno is revising his position. Are you? When the chart the day before was at -2C was there a requirement to cool the water more? |
|
|
|
Post by Andrew on Aug 30, 2013 6:49:34 GMT
this is what Numno said was the fate of the energy: Of course no such thing will happen, the phase change energy will go into changing the water into the crystal structure of ice, which will also expand a bit. (And by which way nature chops down northern rocks and mountains chip by chip.)So Numno said the energy will be used to chop down mountains. Do you agree with that Iceskater? Seems Numno is revising his position. Are you? When the chart the day before was at -2C was there a requirement to cool the water more? It should be obvious that neither myself or Numerouno are revising what we said. As a side comment, Numerouno mentioned that expanding ice fractures rock. However, Numerouno has got a bit mixed up. Energy has to go into ice to form water and energy has to come out of water to form ice. The phase change energy gets removed by cooling before ice will form. If a tonne of water was infintessimally above the freezing point and there was no latent heat of fusion then the the tiniest imaginable amount of cooling would freeze the entire tonne of water. Instead to freeze the entire tonne an enormous amount of additional 'cooling' has to be performed. To calculate the latent heat of fusion, where by definition a calorie is the amount of heat required to change 1g of water by one degree, you would cool water with a fridge that was continuously able to produce a relatively constant cooling force. You would begin with water at say 50C and plot a graph of the rate of change of temperature versus time, and you could then easily calculate the number of calories of cooling power available since you have a known number of grammes of water and a known temperature change. Then during the phase change time you would see the same cooling energy per unit of time did nothing to change the temperature and you would know that this amount of time of cooling was directly related to the number of calories required to produce the phase change of liquid to ice conversion. The amount of cooling required to convert the ice would then be stated as the opposite sign amount of the latent heat of fusion of ice. |
|
|
|
Post by Andrew on Aug 30, 2013 7:43:17 GMT
One explanation for a rising temperature each year after ice has begun forming could be that when the air temperature is only slightly below freezing it is impossible for ice to form unless the sea is exceptionally calm. Once the ice formed, the air would then be insulated from the warmth of the water and the air temperature would fall. Once the ice was broken by wave action the air temperature would likely rise.
There could be all manner of things happening to cause an habitual spike if it is true one exists, but the latent heat of fusion cannot be the cause of it.
|
|
|
|
Post by icefisher on Aug 30, 2013 14:03:26 GMT
You mean I really have to take hot water and cool it before it will freeze and release energy? Imagine that!!! LOL!!!! the discussion was about this chart: (the blue line is 0C) As one can see, and it is seen every year the ocean water cools to about -2C.  So regarding the upward blip I said: this is the time of year when you start seeing heat spikes in the atmosphere from the release of water heat energy as the water freezes. (by time of year it meant when its below 0C)(by upward blip we are looking at slightly more than a 1 deg C burst of energy on the DMI data collectors) this is what Numno said was the fate of the energy: Of course no such thing will happen, the phase change energy will go into changing the water into the crystal structure of ice, which will also expand a bit. (And by which way nature chops down northern rocks and mountains chip by chip.)So Numno said the energy will be used to chop down mountains. Do you agree with that Iceskater? Seems Numno is revising his position. Are you? When the chart the day before was at -2C was there a requirement to cool the water more? You are talking in tongues
It should be obvious that neither myself or Numerouno are revising what we said.
As a side comment, Numerouno mentioned that expanding ice fractures rock.
Why is it that every science conversation you are part of becomes so tedious??Numerouno has not expressed the science correctly in that sentence. The phase change energy does not go into changing the water into ice but rather it has to be removed by cooling before ice will form. If a tonne of water was infintessimally above the freezing point and there was no latent heat of fusion then the the tiniest imaginable amount of cooling would freeze the entire tonne of water. Instead to freeze the entire tonne an enormous amount of additional 'cooling' has to be performed. Well not exactly! Close but you talk about this very thin line for a freezing point that simply does not exist. If you want to be more correct, its correct to say the "melting point" of water is 0C. Water can but does not necessarily freeze at 0C and won't freeze at .00001C. However, almost all water supercools before freezing. This is due to a Savante Arhennius discovery of the activation energy of homogenous nucleation. In the absence of impurities and at sealevel temperature it has been found that water can be cooled to below -40C without freezing. If pressure is increased the freezing point can be further depressed (-74C) This effect is different than water that has a lower freezing point because of impurities like saltwater. That effect is called freezing point depression. But this depression for seawater may not prevent the water temp from jumping up to several degrees upon freezing due to the salts being removed as part of the freezing process as I have not found anything to suggest it does. There also has been a discovery that water goes into some kind of mysterious 3rd state between its melting and freezing states. The ejection of energy from a freezing drop of water has been photographed.rogerjcheng.com/STAGE%20ONE-FREEZING%20of%20A%20WATER%20DROP.htmThe water which was supercooled immediately jumped to 0C upon initiation of freezing and it ejected microdrops of water from its surface. These microdrops held a positive energy charge leaving the drop of water with a negative charge. Science is still learning about the mysterious processes of water. There is quite a few papers out looking at this process as a means of creating thunderstorms. YEA! you want to see energy bursting out of freezing water? YEA! www.igsoc.org/journal.old/8/53/igs_journal_vol08_issue053_pg301-309.pdfIt appears that indeed, since the melting point is not subject to modification, the drop cannot rise above the 0C level without terminating freezing. But it appears it does have some form of exothermic ability to eject heat, perhaps through sublimation, beyond the cooling rate that its temperature mandates. Of course there is no claim here though that such an ejection of heat is necessary to protect crops. The fact that the freezing process itself appears to warm the water up to the melting point of water and increase its cooling ability as heat bursts out of the H20 molecules. This effect can therefore warm anything that is below 0C, like maybe the atmosphere in contact with it. Now does that mean all water super cools? No it doesn't because of the concept of heterogeneous nucleation. 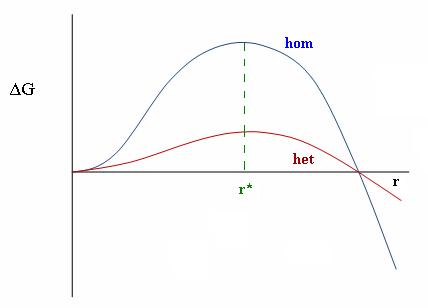 As we can see heterogeneous nucleation reduces the supercooling required to initiate freezing. I suppose its some kind of statistical argument as to whether freezing can occur at all at 0C. It remains a possibility but perhaps a remote one because by far most of the substance in tap water is in fact water and not impurities. Interesing stuff, much more detail than the general concept I had of this before this conversation began. |
|