|
|
Post by nautonnier on Sept 1, 2019 10:51:27 GMT
I think you may be right. But as you will see everyone is far happier talking about applying heat to break bonds, but not as you say where this heat is going and how it stays 'stored' in a free water molecule to be released on condensation. It is not a trivial amount of heat either. In my sorta proto understanding of this, i think stored is maybe the wrong way of describing it in relation to the molecule bonds. During phase change the molecules are taking or releasing the energy required to accomplish the bond requirements from the surrounding environment. (Edit to come....) Yes, so I have a free floating H 2O molecule it has kinetic energy and no nearby other H 2O molecules. It gets attracted to a small particle a 'condensation nucleus' and along with other H 2O molecules a water drop is created. Their kinetic energy is transferred to the condensation nucleus - but where does the latent heat of condensation 'appear from' and go to? It is not small at around 2500 joules/gram |
|
|
|
Post by acidohm on Sept 1, 2019 11:32:19 GMT
In my sorta proto understanding of this, i think stored is maybe the wrong way of describing it in relation to the molecule bonds. During phase change the molecules are taking or releasing the energy required to accomplish the bond requirements from the surrounding environment. (Edit to come....) Yes, so I have a free floating H 2O molecule it has kinetic energy and no nearby other H 2O molecules. It gets attracted to a small particle a 'condensation nucleus' and along with other H 2O molecules a water drop is created. Their kinetic energy is transferred to the condensation nucleus - but where does the latent heat of condensation 'appear from' and go to? It is not small at around 2500 joules/gram Sorry Naut, i had to examine issue further mid post....re-visit quoted post, i added more..... |
|
|
|
Post by Ratty on Sept 1, 2019 12:42:01 GMT
Please ignore me but ...... from DMI. Note how the apparent early re-freeze has kept the temperature 'high' - there is something latent somewhere: 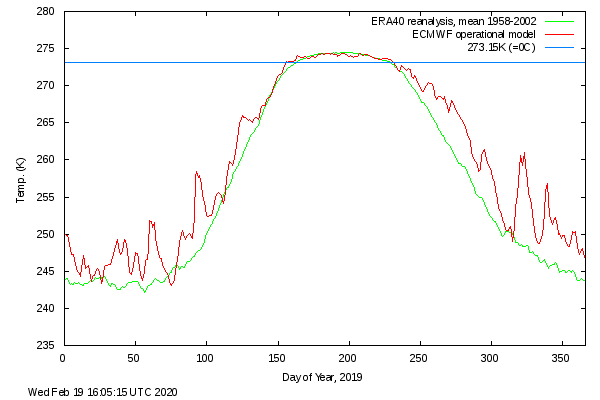 PS: Can you please get to the bottom of this ... and quickly. I'm seventy-four and just had the grandchildren visiting for a few days. No pressure.  |
|
|
|
Post by acidohm on Sept 1, 2019 13:14:29 GMT
Please ignore me but ...... from DMI. Note how the apparent early re-freeze has kept the temperature 'high' - there is something latent somewhere:  PS: Can you please get to the bottom of this ... and quickly. I'm seventy-four and just has the grandchildren visiting for a few days. No pressure.  Im just about to drink beer Ratty, is there time to pass judgement till tommorrow?? I feel this is an important one 😱😆 |
|
|
|
Post by Ratty on Sept 1, 2019 13:17:40 GMT
[ Snip ] Im just about to drink beer Ratty, is there time to pass judgement till tommorrow?? I feel this is an important one 😱😆 OK. Tomorrow is answer time. More beer? Really?  |
|
|
|
Post by acidohm on Sept 1, 2019 13:58:47 GMT
[ Snip ] Im just about to drink beer Ratty, is there time to pass judgement till tommorrow?? I feel this is an important one 😱😆 OK. Tomorrow is answer time. More beer? Really?  Theres so much out there....be criminal to waste it 😯 |
|
|
|
Post by nautonnier on Sept 1, 2019 13:59:03 GMT
Yes, so I have a free floating H 2O molecule it has kinetic energy and no nearby other H 2O molecules. It gets attracted to a small particle a 'condensation nucleus' and along with other H 2O molecules a water drop is created. Their kinetic energy is transferred to the condensation nucleus - but where does the latent heat of condensation 'appear from' and go to? It is not small at around 2500 joules/gram Sorry Naut, i had to examine issue further mid post....re-visit quoted post, i added more..... added text Well OK but where does that energy go? An H2O molecule hits a set of other molecules that kinetic energy would _stop_ the condensation wouldn't it - like the cue ball hitting the triangle of balls. Something makes it pull to a halt but not cause the droplet to break up and that kinetic energy needs to go somewhere how? It would be nice to find a research paper that avoided the hand waving phrases like 'latent heat is released' and actually detailed what happens. |
|
|
|
Post by nautonnier on Sept 1, 2019 14:01:24 GMT
[ Snip ] Im just about to drink beer Ratty, is there time to pass judgement till tommorrow?? I feel this is an important one 😱😆 OK. Tomorrow is answer time. More beer? Really?  Always time for beer - just finished bottling some real ginger beer as in alcoholic, and now I have to bottle another 5 gallons of trappiste style beer to release some containers for drinking water if the hurricane stops the pumped mains water supply.  |
|
|
|
Post by acidohm on Sept 1, 2019 15:00:56 GMT
OK. Tomorrow is answer time. More beer? Really?  Always time for beer - just finished bottling some real ginger beer as in alcoholic, and now I have to bottle another 5 gallons of trappiste style beer to release some containers for drinking water if the hurricane stops the pumped mains water supply.  Good idea, that way if you run out of stored water you could always survive on beer 👍👍 |
|
|
|
Post by nautonnier on Sept 1, 2019 16:21:13 GMT
Always time for beer - just finished bottling some real ginger beer as in alcoholic, and now I have to bottle another 5 gallons of trappiste style beer to release some containers for drinking water if the hurricane stops the pumped mains water supply.  Good idea, that way if you run out of stored water you could always survive on beer 👍👍 The brewed ginger beer would be rather like the wine in the series porridge with Ronnie Barker: “I’d like to warn you, gentlemen, that this should be sipped delicately like a fine liqueur. It shouldn’t be gulped down by the mugful. If you do that you will lose the flavour and the bouquet. You will also lose your power of speech” |
|
|
|
Post by acidohm on Sept 1, 2019 17:22:29 GMT
Good idea, that way if you run out of stored water you could always survive on beer 👍👍 The brewed ginger beer would be rather like the wine in the series porridge with Ronnie Barker: “I’d like to warn you, gentlemen, that this should be sipped delicately like a fine liqueur. It shouldn’t be gulped down by the mugful. If you do that you will lose the flavour and the bouquet. You will also lose your power of speech”I found Sierra Nevada Torpedo in a shop yesterday, i gleefully bought 4 and slurped through the evening, they're 7.2 but maintained power of speech so guess your brew is stronger!?? |
|
|
|
Post by duwayne on Sept 1, 2019 19:27:24 GMT
While not wishing to rekindle arguments on the 'release of latent heat' ..... From the article: "Christy compared the average model projections since 1979 to the most reliable observations — those made by satellites and weather balloons over the vast tropics. The result? In the upper levels of the lower atmosphere, the models predicted seven times as much warming as has been observed." This is the so called missing tropospheric 'hot spot' shown in _all_ the models. Now why would all the models show an increase in _temperature_ (a hot spot)? I believe that the reason is the assumption that has been often stated here that latent heat released by change of state of water is transferred to the other gases in the atmosphere as _sensible heat_ in other words when latent heat is released the surrounding atmosphere is heated up. This does happen (otherwise Dorian would not be 'barreling' (sic) this way now), but sensible heat transfer is not to the level the models expect as there is no hot spot. Now if a large proportion of the latent heat was radiated away as infrared (as shown by the satellites) there would be no temperature change as radiation is not heat, and there would be no hot spot which is what is shown by observation. I will cross post this to Temperature and CO2 to avoid thread drift here  Any comments on the above hypothesis? Nautonnier, you say "Now if a large proportion of the latent heat was radiated away as infrared (as shown by the satellites)...". As I said on the Latent Heat thread, that is not what the satellite imagery shows. It shows temperatures, not quantities of IR emissions. Here is a quote from a Penn State educational module. "(Satellite) infrared imagery is the display of either cloud-top temperatures or Earth's surface temperature (if no clouds are present). www.e-education.psu.edu/meteo3/l5_p5.htmlHere is a longer quote from the article. Some charts are missing but they can be seen by linking to the article. Hopefully you will read through the entire quote so we don't have to come back to this issue again. "Before we delve into what we can learn from infrared satellite imagery, we need to discuss what an infrared satellite image is actually displaying. Just like visible images, infrared images are captured by a radiometer tuned to a specific wavelength. Returning to our atmospheric absorption chart, we see that between roughly 10 microns and 13 microns, there's very little absorption of infrared radiation by the atmosphere. In other words, infrared radiation at these wavelengths emitted by the earth's surface, or by other objects like clouds, gets transmitted to the satellite with very little absorption along the way. You may recall from our previous lesson on radiation that the amount of radiation an object emits is tied to its temperature. Warmer objects emit more radiation than colder objects. So, using the mathematics behind the laws of radiation, computers can convert the amount of infrared radiation received by the satellite to a temperature (formally called a "brightness temperature" even though it has nothing to do with how bright an object looks to human eyes). Finally, these temperatures are converted to a shade of gray or white (or a color, as you're about to see), to create an infrared satellite image. Conventionally, lower temperatures are represented by brighter shades of gray and white, while higher temperatures are represented by darker shades of gray. While visible satellite images pretty much all look the same, that's not the case with infrared images (see the montage of images below). Some infrared images are in grayscale so that they resemble visible images (upper-left), while others include all the colors of the rainbow! Such infrared images that contain different color schemes are usually called enhanced infrared images, not because they are better, but because the color scheme highlights some particular feature on the image (usually very low temperatures). There's really no fundamental difference between a "regular" (grayscale) infrared image and an enhanced infrared image; the coloring does not change the data it is presenting. The key with any IR image is to locate the temperature-color scale (usually on the side or bottom of the image) and match the shading to whatever feature you're looking at. Here are the uncropped images for the "traditional" IR image and lower-right "enhanced image", for reference. Four different views (enhancements) of the same IR image. Four corresponding infrared satellite images with differing color schemes. The "traditional" infrared image is shown in the upper-left. The other satellite images are considered "enhanced" infrared images because they contain colors that mark certain key temperature ranges (in this case very low temperatures). So, we know that an infrared radiometer aboard a satellite measures the intensity of radiation and converts it to a temperature, but what temperature are we measuring? Well, because atmospheric gases don't absorb much radiation between about 10 microns and 13 microns, infrared radiation at these wavelengths mostly gets a "free pass" through the clear air. This means that for a cloudless sky, we are simply seeing the temperature of the earth's surface. To see what I mean, check out this loop of infrared images of the Sahara Desert. Note the very dramatic changes in ground temperatures from night (light gray ground) to day (dark gray/black ground). This is because surface temperatures often dramatically change during the day over deserts, where the broiling sun bakes the earth's surface by day. At night, however, the desert floor often cools off rapidly after sunset. Of course, sometimes clouds block the satellite's view of the surface; so what's being displayed in cloudy areas? Well, while atmospheric gases absorb very little infrared radiation at these wavelengths (and thus emit very little by Kirchhoff's Law), that's not the case for liquid water and ice, which emit very efficiently at these wavelengths. Therefore, any clouds that are in the view of the satellite will be emitting infrared radiation consistent with their temperatures. Furthermore, infrared emitted by the earth's surface is completely absorbed by the clouds above it. Remember that since clouds emit infrared radiation effectively at this wavelength, they also absorb radiation very effectively. So even though there is plenty of infrared radiation coming from below the cloud and even from within the cloud itself, the only radiation that reaches the satellite is from the cloud top. Therefore, infrared imagery is the display of either cloud-top temperatures or Earth's surface temperature (if no clouds are present).So, your hypothesis is based on a misinterpretation of an imagery chart and I can't agree with it. |
|
|
|
Post by nautonnier on Sept 1, 2019 19:57:55 GMT
In that case Duwayne you will be able to explain where and how the latent heat from the condensing/freezing water goes. It does NOT heat the upper troposphere as is expected from sensible heat as there is no tropospheric hot spot.
While I agree that the infrared sensor output has been 'adjusted' to what the climate scientists think it should be.
So the conversion is based on an assumption.
However, satellite imagery DOES show infrared being radiated. Infrared does not have a temperature it is radiation. Perhaps their assumptions on the 'temperature' are based on the non-existent hot spot.
|
|
|
|
Post by nautonnier on Sept 3, 2019 11:58:28 GMT
|
|
|
|
Post by duwayne on Sept 3, 2019 14:17:29 GMT
Nautonnier, do you believe this? |
|