|
|
Post by Belushi TD on Jan 23, 2009 0:30:54 GMT
Socold -
If we're dealing with a true exponential f(x), then a doubling with have half the effect of the previous one... To whit:
If one chicken is funny, and two chickens is half again as funny, then an infinite number of chickens is as funny as 2*one chicken....
Math is a wonderful thing.
1 + 1/2 + 1/4 + 1/8 ...... + 1/2^infinity = 2
There's a finite maximum of energy that CO2 can retain.
Belushi TD
|
|
|
|
Post by socold on Jan 23, 2009 1:19:05 GMT
Socold - If we're dealing with a true exponential f(x), then a doubling with have half the effect of the previous one... To whit: If one chicken is funny, and two chickens is half again as funny, then an infinite number of chickens is as funny as 2*one chicken.... Math is a wonderful thing. 1 + 1/2 + 1/4 + 1/8 ...... + 1/2^infinity = 2 There's a finite maximum of energy that CO2 can retain. Belushi TD Are you even being serious? |
|
|
|
Post by magellan on Jan 23, 2009 3:05:15 GMT
Doubling CO2 will have half the impact of the previous doubling which was half the previous doubling. (I'll leave aside my contention that the CO2 absorption band is already saturated). You have applied the diminishing returns once too many! It's each doubling has the same effect, not each doubling has half the effect of the last. You are confusing the average residence time of a single molecule, which is 2-5 years, with the time it takes for elevated co2 levels to return back to their previous level (which is a logarithmic decay function which still leaves some in the atmosphere thousands of years later. 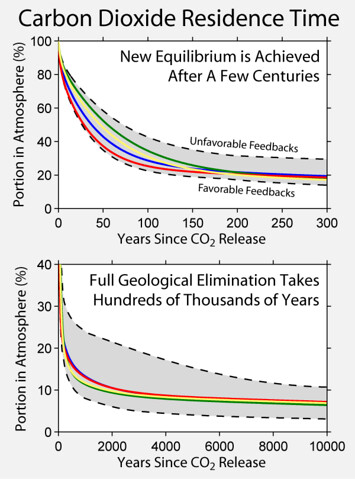 The species living in those periods were by definition adapted to it. Today's species are not necessarily, so you change conditions too fast and they will not have time to evolve an adaptation. It's all about the rapid speed of the change. Did they ever find the missing CO2 sink? |
|
|
|
Post by Belushi TD on Jan 23, 2009 22:41:10 GMT
Yes and no... The chicken bit was intended as humor.
The math was intended to be serious...
Belushi TD
|
|
|
|
Post by trbixler on Jan 24, 2009 1:18:01 GMT
Nice graph but could it be replotted with the Y being the % of atmosphere? ;D Sometimes I think we lose perspective.
|
|
|
|
Post by Acolyte on Jan 24, 2009 2:06:12 GMT
I'm a little puzzled as to how, firstly, anyone can tell how long any given molecule of CO2 remains in the atmosphere & secondly, given that we can talk about it statistically, how, if a molecule, any given molecule, released into atmosphere will be taken up again in 2 - 5 years, it can be said that the ongoing effects last longer.
If CO2 has a 2 - 5 year cycle, or a 12 years cycle, then once that molecule is back in the system the effects cease. Unless I am missing something, to agree that the molecules have a 'shelf life' of so short a time then claim their effects last centrueis or millenia is a little ludicrous.
|
|
|
|
Post by poitsplace on Jan 24, 2009 5:39:52 GMT
I'm a little puzzled as to how, firstly, anyone can tell how long any given molecule of CO 2 remains in the atmosphere & secondly, given that we can talk about it statistically, how, if a molecule, any given molecule, released into atmosphere will be taken up again in 2 - 5 years, it can be said that the ongoing effects last longer. If CO 2 has a 2 - 5 year cycle, or a 12 years cycle, then once that molecule is back in the system the effects cease. Unless I am missing something, to agree that the molecules have a 'shelf life' of so short a time then claim their effects last centrueis or millenia is a little ludicrous. It's really about the state of equilibrium maintained (or lack of it). Talking about individual molecules is just a gross oversimplification to make it easier to understand. When the oceans warm and degas, more CO2 tends to leave the oceans than is taken in. When the oceans cool and solubility increases they start taking in CO2. It may in fact be possible for the oceans to absorb CO2 as fast as humans are making it if the temperature drops. Actually the proposed "feedbacks" for CO2 operate in much the same way...they care little where the equilibrium shift comes from, only the level. If solar forcing, or the current lack of it, overpowers CO2 (probably will) then they kick into a mode that amplifies cooling. Ironically if the temperature falls CO2 actually turns into more of a feedback. Remember, equilibrium shifts are about balance. With more CO2 in the air the ocean's ability to dissolve it go up. The partial pressure of CO2 has increased. Just like injecting CO2 into a soft drink bottle full of distilled water...higher pressures of the gas over the water mean more of the gas can be dissolved in the water. If the temperature drops to 1970's levels or lower CO2 may begin to fall. The cooling has only just started. hmmm, this is probably not the information you wanted but...what the heck. |
|
|
|
Post by Acolyte on Jan 24, 2009 8:12:36 GMT
It's interesting though... What I'm trying to work out is this; socold said: You are confusing the average residence time of a single molecule, which is 2-5 years, with the time it takes for elevated co2 levels to return back to their previous level (which is a logarithmic decay function which still leaves some in the atmosphere thousands of years later. If CO 2 residence time is 2 - 5 years, or even 12 years as I've seen research about, how can it take longer for elevated levels to return to previous levels. It would seem that the implication of the above statement is of something new in the discussion, or maybe magic. We have an event that comes along, remains for a short time but when it goes away the elevated levels caused by that event stay behind. Doesn't seem to make sense. Current level CO 2 = 200ppm, Added is 35ppm so for somewhere between 2 & 12 years the level is 235ppm. Now, at the end of that period the level returns to 200ppm. Now I realise I'm picking a moment in time here but the ongoing rise will still, in between 2 & 12 years revert back. So it seems to me that any ongoing rise must come from an ever increasing rise in CO 2 so for agw to reach levels predicted by models we'd need an ever-increasing release of CO 2 which, given current energy & oil situation, seems patently impossible. So am I missing something...? |
|
|
|
Post by nautonnier on Jan 24, 2009 10:19:09 GMT
I'm a little puzzled as to how, firstly, anyone can tell how long any given molecule of CO 2 remains in the atmosphere & secondly, given that we can talk about it statistically, how, if a molecule, any given molecule, released into atmosphere will be taken up again in 2 - 5 years, it can be said that the ongoing effects last longer. If CO 2 has a 2 - 5 year cycle, or a 12 years cycle, then once that molecule is back in the system the effects cease. Unless I am missing something, to agree that the molecules have a 'shelf life' of so short a time then claim their effects last centrueis or millenia is a little ludicrous. It's really about the state of equilibrium maintained (or lack of it). Talking about individual molecules is just a gross oversimplification to make it easier to understand. When the oceans warm and degas, more CO2 tends to leave the oceans than is taken in. When the oceans cool and solubility increases they start taking in CO2. It may in fact be possible for the oceans to absorb CO2 as fast as humans are making it if the temperature drops. Actually the proposed "feedbacks" for CO2 operate in much the same way...they care little where the equilibrium shift comes from, only the level. If solar forcing, or the current lack of it, overpowers CO2 (probably will) then they kick into a mode that amplifies cooling. Ironically if the temperature falls CO2 actually turns into more of a feedback. Remember, equilibrium shifts are about balance. With more CO2 in the air the ocean's ability to dissolve it go up. The partial pressure of CO2 has increased. Just like injecting CO2 into a soft drink bottle full of distilled water...higher pressures of the gas over the water mean more of the gas can be dissolved in the water. If the temperature drops to 1970's levels or lower CO2 may begin to fall. The cooling has only just started. hmmm, this is probably not the information you wanted but...what the heck. . I think you are right - CO2 is merely an indicator of Ocean temperatures. It has a minor radiative forcing effect but that is already saturated. So what we are seeing in the current CO2 rise is the effect of the oceans warming. If the oceans cool then CO2 levels will start to decrease - regardless of how many coal powered power stations there are in China. |
|
|
|
Post by socold on Jan 24, 2009 15:07:39 GMT
For example take a hypothetical atmosphere with 2000 billion tons of co2 and each year 700 billion tons are added and 700 billion removed and where the lifetime for a molecule of co2 in the atmosphere is less than 10 years. The atmosphere stays at 2000 billion tons because co2 in = co2 out.
If you suddenly add a 500 billion ton blob of co2 to the atmosphere then now the atmosphere has 2500 billion tons of co2. How long will it take for that extra 500 billion tons to be removed? If the co2 added and removed each year stays at 700 billion tons then the atmosphere will stay at 2500 and that 500 billion blob will never be removed. Even though the lifetime for a molecule of co2 is still less than 10 years.
However in reality if you suddenly up the amount of co2 in the atmosphere then you increase absorption slightly. So lets say absorption increases from 700 billion tons per year to 720 billion tons. That means the atmosphere will lose 20 billion tons of co2 net each year. At that rate it will take about 25 years to remove all the extra 500 billion tons.
Except that as the co2 in the atmosphere does fall, the absorption will decrease. Perhaps after a decade atmospheric co2 level is at 2300 billion tons, but now absorption is now just 710 billion tons per years. At that rate it means it will take a further 30 years to remove all of the remaining 300 billion tons of co2.
But after 10 more years when co2 level is at about 2200 billion tons, absorption has dropped further to now just 705 billion tons per year, so it will take a further 40 years at this rate to remove all the remaining extra co2. This means the co2 fall follows an exponential decay curve.
|
|
|
|
Post by poitsplace on Jan 24, 2009 16:59:13 GMT
Except that as the co2 in the atmosphere does fall, the absorption will decrease. Perhaps after a decade atmospheric co2 level is at 2300 billion tons, but now absorption is now just 710 billion tons per years. At that rate it means it will take a further 30 years to remove all of the remaining 300 billion tons of co2. But after 10 more years when co2 level is at about 2200 billion tons, absorption has dropped further to now just 705 billion tons per year, so it will take a further 40 years at this rate to remove all the remaining extra co2. This means the co2 fall follows an exponential decay curve. We're both on the same page here actually. I was actually referring to the double whammy of temperature AND increased levels. We have no idea what the uptake rate of the oceans are at cooler temperatures AND higher concentrations. What we DO know is that the oceans, weighing in at a whopping 1.4 sextillion kilograms, could EASILY absorb the entire yearly CO2 output of mankind for centuries given half a chance. The real question is...at what temperature/concentration will the yearly uptake equal man's output? I think it's quite possible that given the amount the CO2 increase varies each year due to temperature... if indeed we did drop below 1970's temperatures...CO2 increases might actually stop or potentially fall. Again, I'm not saying fall back to pre-industrial levels...just not rise or lower slightly from current levels. |
|
|
|
Post by socold on Jan 24, 2009 19:29:36 GMT
yea i agree, if it gets cold enough then co2 rise should stall or even reverse.
|
|
|
|
Post by nautonnier on Jan 24, 2009 20:54:11 GMT
For example take a hypothetical atmosphere with 2000 billion tons of co2 and each year 700 billion tons are added and 700 billion removed and where the lifetime for a molecule of co2 in the atmosphere is less than 10 years. The atmosphere stays at 2000 billion tons because co2 in = co2 out. If you suddenly add a 500 billion ton blob of co2 to the atmosphere then now the atmosphere has 2500 billion tons of co2. How long will it take for that extra 500 billion tons to be removed? If the co2 added and removed each year stays at 700 billion tons then the atmosphere will stay at 2500 and that 500 billion blob will never be removed. Even though the lifetime for a molecule of co2 is still less than 10 years. However in reality if you suddenly up the amount of co2 in the atmosphere then you increase absorption slightly. So lets say absorption increases from 700 billion tons per year to 720 billion tons. That means the atmosphere will lose 20 billion tons of co2 net each year. At that rate it will take about 25 years to remove all the extra 500 billion tons. Except that as the co2 in the atmosphere does fall, the absorption will decrease. Perhaps after a decade atmospheric co2 level is at 2300 billion tons, but now absorption is now just 710 billion tons per years. At that rate it means it will take a further 30 years to remove all of the remaining 300 billion tons of co2. But after 10 more years when co2 level is at about 2200 billion tons, absorption has dropped further to now just 705 billion tons per year, so it will take a further 40 years at this rate to remove all the remaining extra co2. This means the co2 fall follows an exponential decay curve. You should talk to a market gardener. In actuality there would be a significant increase in worldwide plant fecundity that would start significantly increasing the uptake of CO 2. www.advancegreenhouses.com/use_of_co2_in_a_greenhouse.htm"The best time to add CO2 is from dawn to dusk. CO2 ranges from 400 to 500 ppm during the night due to plant respiration. Right after sunrise a level will drop to about 300 ppm. After three to four hours of early sun light it will drop to 100 to 250 ppm at which time growth will stop. If you add CO2 during the winter months when ventilators are closed and CO2 concentrations are low, you will get increased yield and bloom which normally happens during the spring and summer.
This is a formula which can be used to figure what amount of CO2 must be added to reach 1000 ppm.
9 ft.³ of CO2 per hour per 1000 ft.² yields 1000 ppm"Note at 250ppm GROWTH WILL STOP - Yet the 'preindustrial atmospheric concentration of 1750AD is quoted as 275ppm - so very close to the edge of plant growth stopping!! Think of that when you are having your 5 portions of fruit and veg and are talking of trying to return the world to only 25ppm above plant growth stopping. The increased uptake of CO 2 may already be happening since many of the 1970's doom-laden forecasts of starvation have not happened despite massive increases in population so food production has, until now, been able to keep pace with all those extra mouths. Things in nature are not 'simple' balances - putting more CO 2 in the air will not only be a physical input output balance positive and negative feedbacks including plants and ocean algae will immediately kick in. BTW calculate the amount of fossil fuel of your 500billion ton 'blob' of CO 2 then work out how LONG it would take to generate that amount and see if the usable reserves match that amount. Then add the increase in biomass due to uptake of the CO 2 see what answer you get then  Include the additional food crops that will be available. |
|
|
|
Post by Acolyte on Jan 25, 2009 0:19:02 GMT
For example take a hypothetical atmosphere with 2000 billion tons of co2 and each year 700 billion tons are added and 700 billion removed and where the lifetime for a molecule of co2 in the atmosphere is less than 10 years. The atmosphere stays at 2000 billion tons because co2 in = co2 out. If you suddenly add a 500 billion ton blob of co2 to the atmosphere then now the atmosphere has 2500 billion tons of co2. How long will it take for that extra 500 billion tons to be removed? If the co2 added and removed each year stays at 700 billion tons then the atmosphere will stay at 2500 and that 500 billion blob will never be removed. Even though the lifetime for a molecule of co2 is still less than 10 years. However in reality if you suddenly up the amount of co2 in the atmosphere then you increase absorption slightly. So lets say absorption increases from 700 billion tons per year to 720 billion tons. That means the atmosphere will lose 20 billion tons of co2 net each year. At that rate it will take about 25 years to remove all the extra 500 billion tons. Except that as the co2 in the atmosphere does fall, the absorption will decrease. Perhaps after a decade atmospheric co2 level is at 2300 billion tons, but now absorption is now just 710 billion tons per years. At that rate it means it will take a further 30 years to remove all of the remaining 300 billion tons of co2. But after 10 more years when co2 level is at about 2200 billion tons, absorption has dropped further to now just 705 billion tons per year, so it will take a further 40 years at this rate to remove all the remaining extra co2. This means the co2 fall follows an exponential decay curve. Thanks for the explanation - that makes it clearer. I wasn't factoring in a fixed absorption. But such a fixed rater would have to mean a change in how long any given molecule of CO 2 stays up there wouldn't it? |
|
|
|
Post by socold on Jan 25, 2009 18:01:29 GMT
Yea if the rate was fixed and the amount in the atmosphere increased then the lifetime for a molecule would probably change.
|
|